Intravitreal delivery of AAV8 retinoschisin results in cell type-specific gene expression and retinal rescue in the Rs1-KO mouse
- PMID: 19458650
- PMCID: PMC2774250
- DOI: 10.1038/gt.2009.61
Intravitreal delivery of AAV8 retinoschisin results in cell type-specific gene expression and retinal rescue in the Rs1-KO mouse
Erratum in
- Gene Ther. 2009 Jul;16(7):941
Abstract
X-linked juvenile retinoschisis (XLRS) is a neurodevelopmental abnormality caused by retinoschisin gene mutations. XLRS is characterized by splitting through the retinal layers and impaired synaptic transmission of visual signals resulting in impaired acuity and a propensity to retinal detachment. Several groups have treated murine retinoschisis models successfully using adeno-associated virus (AAV) vectors. Owing to the fragile nature of XLRS retina, translating this therapy to the clinic may require an alternative to invasive subretinal vector administration. Here we show that all layers of the retinoschisin knockout (Rs1-KO) mouse retina can be transduced efficiently with AAV vectors administered by simple vitreous injection. Retinoschisin expression was restricted to the neuroretina using a new vector that uses a 3.5-kb human retinoschisin promoter and an AAV type 8 capsid. Intravitreal administration to Rs1-KO mice resulted in robust retinoschisin expression with a retinal distribution similar to that observed in wild-type retina, including the expression by the photoreceptors lying deep in the retina. No off-target expression was observed. Rs1-KO mice treated with this vector showed a decrease in the schisis cavities and had improved retinal signaling evaluated by recording the electroretinogram 11-15 weeks after the application.
Figures
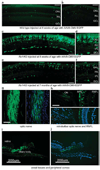

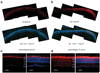



References
-
- Tantri A, Vrabec TR, Cu-Unjieng A, Frost A, Annesley WH, Jr, Donoso LA. X-linked retinoschisis: a clinical and molecular genetic review. Surv Ophthalmol. 2004;49:214–230. - PubMed
-
- Prenner JL, Capone A, Jr, Ciaccia S, Takada Y, Sieving PA, Trese MT. Congenital X-linked retinoschisis classification system. Retina. 2006;26(7 Suppl):S61–S64. - PubMed
-
- Peachey NS, Fishman GA, Derlacki DJ, Brigell MG. Psychophysical and electroretinographic findings in X-linked juvenile retinoschisis. Arch Ophthalmol. 1987;105:513–516. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

