Absence of alpha 4 but not beta 2 integrins restrains development of chronic allergic asthma using mouse genetic models
- PMID: 19463772
- PMCID: PMC3696022
- DOI: 10.1016/j.exphem.2009.03.010
Absence of alpha 4 but not beta 2 integrins restrains development of chronic allergic asthma using mouse genetic models
Abstract
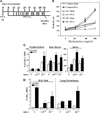
Objective: Chronic asthma is characterized by ongoing recruitment of inflammatory cells and airway hyperresponsiveness leading to structural airway remodeling. Although alpha 4 beta 1 and beta2 integrins regulate leukocyte migration in inflammatory diseases and play decisive roles in acute asthma, their role has not been explored under the chronic asthma setting. To extend our earlier studies with alpha 4(Delta/Delta) and beta2(-/-) mice, which showed that both alpha 4 and beta2 integrins have nonredundant regulatory roles in acute ovalbumin (OVA)-induced asthma, we explored to what extent these molecular pathways control development of structural airway remodeling in chronic asthma.
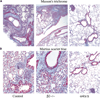
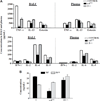
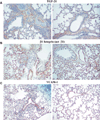
Materials and methods: Control, alpha 4(Delta/Delta), and beta2(-/-) mouse groups, sensitized by intraperitoneal OVA as allergen, received intratracheal OVA periodically over days 8 to 55 to induce a chronic asthma phenotype. Post-OVA assessment of inflammation and pulmonary function (airway hyperresponsiveness), together with airway modeling measured by goblet cell metaplasia, collagen content of lung, and transforming growth factor beta1 expression in lung homogenates, were evaluated.
Results: In contrast to control and beta2(-/-) mice, alpha 4(Delta/Delta) mice failed to develop and maintain the composite chronic asthma phenotype evaluated as mentioned and subepithelial collagen content was comparable to baseline. These data indicate that beta2 integrins, although required for inflammatory migration in acute asthma, are dispensable for structural remodeling in chronic asthma.
Conclusion: alpha 4 integrins appear to have a regulatory role in directing transforming growth factor beta-induced collagen deposition and structural alterations in lung architecture likely through interactions of Th2 cells, eosinophils, or mast cells with endothelium, resident airway cells, and/or extracellular matrix.
Figures


Similar articles
-
ISO-1, a macrophage migration inhibitory factor antagonist, inhibits airway remodeling in a murine model of chronic asthma.Mol Med. 2010 Sep-Oct;16(9-10):400-8. doi: 10.2119/molmed.2009.00128. Epub 2010 May 14. Mol Med. 2010. PMID: 20485865 Free PMC article.
-
Alpha4 and beta2 integrins have nonredundant roles for asthma development, but for optimal allergen sensitization only alpha4 is critical.Exp Hematol. 2007 Apr;35(4):605-17. doi: 10.1016/j.exphem.2007.01.052. Exp Hematol. 2007. PMID: 17379071
-
Experimental protocol for development of adjuvant-free murine chronic model of allergic asthma.J Immunol Methods. 2019 May;468:10-19. doi: 10.1016/j.jim.2019.03.002. Epub 2019 Mar 14. J Immunol Methods. 2019. PMID: 30880263
-
Roles of integrin activation in eosinophil function and the eosinophilic inflammation of asthma.J Leukoc Biol. 2008 Jan;83(1):1-12. doi: 10.1189/jlb.0607344. Epub 2007 Oct 10. J Leukoc Biol. 2008. PMID: 17906117 Free PMC article. Review.
-
Pathobiology of Airway Remodeling in Asthma: The Emerging Role of Integrins.J Asthma Allergy. 2022 May 11;15:595-610. doi: 10.2147/JAA.S267222. eCollection 2022. J Asthma Allergy. 2022. PMID: 35592385 Free PMC article. Review.
Cited by
-
Clinical implications of CD4+ T cell subsets in adult atopic asthma patients.Allergy Asthma Clin Immunol. 2018 Mar 2;14:7. doi: 10.1186/s13223-018-0231-3. eCollection 2018. Allergy Asthma Clin Immunol. 2018. PMID: 29507584 Free PMC article.
-
Anti-IL-5 attenuates activation and surface density of β(2) -integrins on circulating eosinophils after segmental antigen challenge.Clin Exp Allergy. 2013 Mar;43(3):292-303. doi: 10.1111/j.1365-2222.2012.04065.x. Clin Exp Allergy. 2013. PMID: 23414537 Free PMC article.
-
Defining the molecular role of gp91phox in the immune manifestation of acute allergic asthma using a preclinical murine model.Clin Mol Allergy. 2012 Jan 4;10(1):2. doi: 10.1186/1476-7961-10-2. Clin Mol Allergy. 2012. PMID: 22216879 Free PMC article.
-
α(M)β(2) integrin-mediated adhesion and motility of IL-5-stimulated eosinophils on periostin.Am J Respir Cell Mol Biol. 2013 Apr;48(4):503-10. doi: 10.1165/rcmb.2012-0150OC. Am J Respir Cell Mol Biol. 2013. PMID: 23306834 Free PMC article. Clinical Trial.
-
Dissecting asthma pathogenesis through study of patterns of cellular traffic indicative of molecular switches operative in inflammation.Prog Stem Cell. 2015;2(1):1. doi: 10.7603/s40855-015-0001-2. Epub 2015 Dec 25. Prog Stem Cell. 2015. PMID: 27512648 Free PMC article.
References
-
- Wills-Karp M. Immunologic basis of antigen-induced airway hyper-responsiveness. Annu Rev Immunol. 1999;17:255–281. - PubMed
-
- Shum BO, Rolph MS, Sewell WA. Mechanisms in allergic airway inflammation-lessons from studies in the mouse. Expert Rev Mol Med. 2008;10:e15. - PubMed
-
- Kariyawasam HH, Robinson DS. The role of eosinophils in airway tissue remodelling in asthma. Curr Opin Immunol. 2007;19:681–686. - PubMed
-
- Benayoun L, Druilhe A, Dombret M-C, Aubier M, Pretolani M. Airway structural alterations selectively associated with severe asthma. Am J Respir Crit Care Med. 2003;167:1360–1368. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

