Early events, kinetic intermediates and the mechanism of protein folding in cytochrome C
- PMID: 19468320
- PMCID: PMC2680628
- DOI: 10.3390/ijms10041476
Early events, kinetic intermediates and the mechanism of protein folding in cytochrome C
Erratum in
- Int J Mol Sci. 2009 Apr;10(4):1728
Abstract
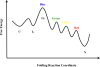
Kinetic studies of the early events in cytochrome c folding are reviewed with a focus on the evidence for folding intermediates on the submillisecond timescale. Evidence from time-resolved absorption, circular dichroism, magnetic circular dichroism, fluorescence energy and electron transfer, small-angle X-ray scattering and amide hydrogen exchange studies on the t < or = 1 ms timescale reveals a picture of cytochrome c folding that starts with the approximately 1-micros conformational diffusion dynamics of the unfolded chains. A fractional population of the unfolded chains collapses on the 1 - 100 micros timescale to a compact intermediate I(C) containing some native-like secondary structure. Although the existence and nature of I(C) as a discrete folding intermediate remains controversial, there is extensive high time-resolution kinetic evidence for the rapid formation of I(C) as a true intermediate, i.e., a metastable state separated from the unfolded state by a discrete free energy barrier. Final folding to the native state takes place on millisecond and longer timescales, depending on the presence of kinetic traps such as heme misligation and proline mis-isomerization. The high folding rates observed in equilibrium molten globule models suggest that I(C) may be a productive folding intermediate. Whether it is an obligatory step on the pathway to the high free energy barrier associated with millisecond timescale folding to the native state, however, remains to be determined.
Keywords: Collapsed intermediate; Trp59 fluorescence; amide hydrogen exchange; conformational diffusion; disordered tertiary structure; far-UV circular dichroism; heme misligation; magnetic circular dichroism; molten globule; secondary structure formation; small-angle X-ray scattering; thermophiles; three-state pathway; time-resolved spectroscopy; unfolded chains.
Figures

Similar articles
-
Folding of horse cytochrome c in the reduced state.J Mol Biol. 2001 Oct 5;312(5):1135-60. doi: 10.1006/jmbi.2001.4993. J Mol Biol. 2001. PMID: 11580255
-
Characterization of a partially unfolded structure of cytochrome c induced by sodium dodecyl sulphate and the kinetics of its refolding.Eur J Biochem. 1998 Jun 15;254(3):662-70. doi: 10.1046/j.1432-1327.1998.2540662.x. Eur J Biochem. 1998. PMID: 9688280
-
The folding kinetics of the SDS-induced molten globule form of reduced cytochrome c.Biochemistry. 2008 May 13;47(19):5450-9. doi: 10.1021/bi702452u. Epub 2008 Apr 17. Biochemistry. 2008. PMID: 18416561
-
Probing early events in ferrous cytochrome c folding with time-resolved natural and magnetic circular dichroism spectroscopies.Curr Protein Pept Sci. 2009 Oct;10(5):464-75. doi: 10.2174/138920309789352001. Curr Protein Pept Sci. 2009. PMID: 19538147 Free PMC article. Review.
-
Protein folding intermediates and pathways studied by hydrogen exchange.Annu Rev Biophys Biomol Struct. 2000;29:213-38. doi: 10.1146/annurev.biophys.29.1.213. Annu Rev Biophys Biomol Struct. 2000. PMID: 10940248 Review.
Cited by
-
Nanosecond time-resolved polarization spectroscopies: tools for probing protein reaction mechanisms.Methods. 2010 Sep;52(1):3-11. doi: 10.1016/j.ymeth.2010.04.015. Epub 2010 May 11. Methods. 2010. PMID: 20438842 Free PMC article.
-
Interconnection of salt-induced hydrophobic compaction and secondary structure formation depends on solution conditions: revisiting early events of protein folding at single molecule resolution.J Biol Chem. 2012 Mar 30;287(14):11546-55. doi: 10.1074/jbc.M111.315648. Epub 2012 Feb 2. J Biol Chem. 2012. PMID: 22303014 Free PMC article.
-
Chlorogenic Acid Enhances the Chaperone Potential of BSA at Physiological Concentrations on Model Protein Cytochrome c.Cell Biochem Biophys. 2025 Mar;83(1):845-856. doi: 10.1007/s12013-024-01516-1. Epub 2024 Sep 22. Cell Biochem Biophys. 2025. PMID: 39306822
-
Heterogeneous folding and stretched kinetics.Proc Natl Acad Sci U S A. 2020 Aug 11;117(32):18915-18917. doi: 10.1073/pnas.2009596117. Epub 2020 Jul 28. Proc Natl Acad Sci U S A. 2020. PMID: 32723817 Free PMC article. No abstract available.
-
Genetically Encoded Fluorescent Probe for Detection of Heme-Induced Conformational Changes in Cytochrome c.Biosensors (Basel). 2023 Sep 18;13(9):890. doi: 10.3390/bios13090890. Biosensors (Basel). 2023. PMID: 37754124 Free PMC article.
References
-
- Pascher T, Chesick JP, Winkler JR, Gray HB. Protein folding triggered by electron transfer. Science. 1996;271:1556–1560. - PubMed
-
- Chan CK, Hofrichter J, Eaton WA, Winkler JR, Gray HB. Optical triggers of protein folding. Science. 1996;274:628–629. - PubMed
-
- Babul J, Stellwagen E. The existence of heme-protein coordinate-covalent bonds in denaturing solvents. Biopolymers. 1971;10:2359–2361. - PubMed
-
- Muthukrishnan K, Nall BT. Effective concentrations of amino acid side chains in an unfolded protein. Biochemistry. 1991;30:4706–4710. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

