Control and regulation of integrated mitochondrial function in metabolic and transport networks
- PMID: 19468321
- PMCID: PMC2680629
- DOI: 10.3390/ijms10041500
Control and regulation of integrated mitochondrial function in metabolic and transport networks
Abstract
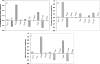
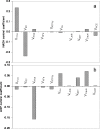
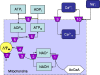
The pattern of flux and concentration control coefficients in an integrated mitochondrial energetics model is examined by applying a generalized matrix method of control analysis to calculate control coefficients, as well as response coefficients The computational model of Cortassa et al. encompasses oxidative phosphorylation, the TCA cycle, and Ca(2+) dynamics. Control of ATP synthesis, TCA cycle, and ANT fluxes were found to be distributed among various mitochondrial processes. Control is shared by processes associated with ATP/ADP production and transport, as well as by Ca(2+) dynamics. The calculation also analyzed the control of the concentrations of key regulatory ions and metabolites (Ca(2+), NADH, ADP). The approach we have used demonstrates how properties of integrated systems may be understood through applications of computational modeling and control analysis.
Keywords: Mitochondrial computational model; calcium dynamics; control by diffuse loops; distributed control; excitation-contraction coupling; metabolic control analysis; mitochondrial energetics.
Figures
Similar articles
-
Metabolic control analysis applied to mitochondrial networks.Annu Int Conf IEEE Eng Med Biol Soc. 2011;2011:4673-6. doi: 10.1109/IEMBS.2011.6091157. Annu Int Conf IEEE Eng Med Biol Soc. 2011. PMID: 22255380 Free PMC article.
-
Numerical modelling of the effects of cold atmospheric plasma on mitochondrial redox homeostasis and energy metabolism.Sci Rep. 2019 Nov 20;9(1):17138. doi: 10.1038/s41598-019-53219-w. Sci Rep. 2019. PMID: 31748630 Free PMC article.
-
Control and regulation of mitochondrial energetics in an integrated model of cardiomyocyte function.Biophys J. 2009 Mar 18;96(6):2466-78. doi: 10.1016/j.bpj.2008.12.3893. Biophys J. 2009. PMID: 19289071 Free PMC article.
-
Role of mitochondrial Ca2+ in the regulation of cellular energetics.Biochemistry. 2012 Apr 10;51(14):2959-73. doi: 10.1021/bi2018909. Epub 2012 Mar 29. Biochemistry. 2012. PMID: 22443365 Free PMC article. Review.
-
Cell energy metabolism: An update.Biochim Biophys Acta Bioenerg. 2020 Nov 1;1861(11):148276. doi: 10.1016/j.bbabio.2020.148276. Epub 2020 Jul 24. Biochim Biophys Acta Bioenerg. 2020. PMID: 32717222 Review.
Cited by
-
Modeling Krebs cycle from liver, heart and hepatoma mitochondria, supported Complex I as target for specific inhibition of cancer cell proliferation.Front Oncol. 2025 Mar 26;15:1557638. doi: 10.3389/fonc.2025.1557638. eCollection 2025. Front Oncol. 2025. PMID: 40206582 Free PMC article.
-
Intrinsic aerobic capacity correlates with greater inherent mitochondrial oxidative and H2O2 emission capacities without major shifts in myosin heavy chain isoform.J Appl Physiol (1985). 2012 Nov;113(10):1624-34. doi: 10.1152/japplphysiol.01475.2011. Epub 2012 Sep 20. J Appl Physiol (1985). 2012. PMID: 22995392 Free PMC article.
-
Mitochondrial network energetics in the heart.Wiley Interdiscip Rev Syst Biol Med. 2012 Nov-Dec;4(6):599-613. doi: 10.1002/wsbm.1188. Epub 2012 Aug 15. Wiley Interdiscip Rev Syst Biol Med. 2012. PMID: 22899654 Free PMC article. Review.
-
Mitochondrial recoupling: a novel therapeutic strategy for cancer?Br J Cancer. 2011 Aug 9;105(4):469-74. doi: 10.1038/bjc.2011.245. Epub 2011 Jun 28. Br J Cancer. 2011. PMID: 21712825 Free PMC article. Review.
-
Modular organization of cardiac energy metabolism: energy conversion, transfer and feedback regulation.Acta Physiol (Oxf). 2015 Jan;213(1):84-106. doi: 10.1111/apha.12287. Epub 2014 Apr 18. Acta Physiol (Oxf). 2015. PMID: 24666671 Free PMC article. Review.
References
-
- Aon MA, Cortassa S.Metabolic dynamics in cells viewed as multilayered, distributed, mass-energy-information networks Encyclopedia of Genetics, Genomics, Proteomics and Bioinformatics Jorde L, Little P, Dunn M, Subramaniam S.Eds; John Wiley & Sons Inc; New York, USA: 20063
-
- Saks V, Dzeja PP, Guzun R, Aliev MK, Vendelin M, Terzic A, Wallimann T.System analysis of cardiac energetics-excitation-contraction coupling: Integration of mitochondrial respiration, phosphotransfer pathways, metabolic pacing, and substrate supply in the heart Molecular System Bioenergetics Energy for Life Saks V.Ed; Wiley-VCH Verlag GmbH&Co. KGaA; Weinheim, Darmstadt, Germany: 2007367–405.
-
- Kacser H, Burns JA. The control of flux. Symp. Soc. Exp. Biol. 1973;27:65–104. - PubMed
-
- Heinrich R, Rapoport TA. A linear steady-state treatment of enzymatic chains. General properties, control and effector strength. Eur. J. Biochem. 1974;42:89–95. - PubMed
-
- Higgins J.Dynamics and control in cellular reactions Control of Energy Metabolism Chance B, Estabrook RW, Williamson JR.Eds; Academic Press; New York, USA: 196513–46.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

