A distinct pool of phosphatidylinositol 4,5-bisphosphate in caveolae revealed by a nanoscale labeling technique
- PMID: 19470488
- PMCID: PMC2695096
- DOI: 10.1073/pnas.0900216106
A distinct pool of phosphatidylinositol 4,5-bisphosphate in caveolae revealed by a nanoscale labeling technique
Erratum in
- Proc Natl Acad Sci U S A. 2009 Jul 14;106(28):11818
Abstract
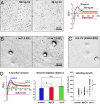
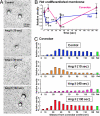
Multiple functionally independent pools of phosphatidylinositol 4,5-bisphosphate [PI(4,5)P(2)] have been postulated to occur in the cell membrane, but the existing techniques lack sufficient resolution to unequivocally confirm their presence. To analyze the distribution of PI(4,5)P(2) at the nanoscale, we developed an electron microscopic technique that probes the freeze-fractured membrane preparation by the pleckstrin homology domain of phospholipase C-delta1. This method does not require chemical fixation or expression of artificial probes, it is applicable to any cell in vivo and in vitro, and it can define the PI(4,5)P(2) distribution quantitatively. By using this method, we found that PI(4,5)P(2) is highly concentrated at the rim of caveolae both in cultured fibroblasts and mouse smooth muscle cells in vivo. PI(4,5)P(2) was also enriched in the coated pit, but only a low level of clustering was observed in the flat undifferentiated membrane. When cells were treated with angiotensin II, the PI(4,5)P(2) level in the undifferentiated membrane decreased to 37.9% within 10 sec and then returned to the initial level. Notably, the PI(4,5)P(2) level in caveolae showed a slower but more drastic change and decreased to 20.6% at 40 sec, whereas the PI(4,5)P(2) level in the coated pit was relatively constant and decreased only to 70.2% at 10 sec. These results show the presence of distinct PI(4,5)P(2) pools in the cell membrane and suggest a unique role for caveolae in phosphoinositide signaling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Di Paolo G, De Camilli P. Phosphoinositides in cell regulation and membrane dynamics. Nature. 2006;443:651–657. - PubMed
-
- McLaughlin S, Wang J, Gambhir A, Murray D. PIP2 and proteins: Interactions, organization, and information flow. Annu Rev Biophys Biomol Struct. 2002;31:151–175. - PubMed
-
- Janmey PA, Lindberg U. Cytoskeletal regulation: Rich in lipids. Nat Rev Mol Cell Biol. 2004;5:658–666. - PubMed
-
- McLaughlin S, Murray D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature. 2005;438:605–611. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

