A novel sulindac derivative that does not inhibit cyclooxygenases but potently inhibits colon tumor cell growth and induces apoptosis with antitumor activity
- PMID: 19470791
- PMCID: PMC3227417
- DOI: 10.1158/1940-6207.CAPR-09-0001
A novel sulindac derivative that does not inhibit cyclooxygenases but potently inhibits colon tumor cell growth and induces apoptosis with antitumor activity
Abstract
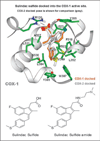
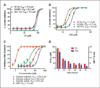
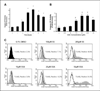
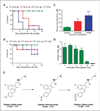
Nonsteroidal anti-inflammatory drugs such as sulindac have shown promising antineoplastic activity, although toxicity from cyclooxygenase (COX) inhibition and the suppression of prostaglandin synthesis limits their use for chemoprevention. Previous studies have concluded that the mechanism responsible for their antineoplastic activity may be COX independent. To selectively design out the COX inhibitory activity of sulindac sulfide (SS), in silico modeling studies were done that revealed the crucial role of the carboxylate moiety for COX-1 and COX-2 binding. These studies prompted the synthesis of a series of SS derivatives with carboxylate modifications that were screened for tumor cell growth and COX inhibitory activity. A SS amide (SSA) with a N,N-dimethylethyl amine substitution was found to lack COX-1 and COX-2 inhibitory activity, yet potently inhibit the growth of human colon tumor cell lines, HT-29, SW480, and HCT116 with IC(50) values of 2 to 5 micromol/L compared with 73 to 85 micromol/L for SS. The mechanism of growth inhibition involved the suppression of DNA synthesis and apoptosis induction. Oral administration of SSA was well-tolerated in mice and generated plasma levels that exceeded its in vitro IC(50) for tumor growth inhibition. In the human HT-29 colon tumor xenograft mouse model, SSA significantly inhibited tumor growth at a dosage of 250 mg/kg. Combined treatment of SSA with the chemotherapeutic drug, Camptosar, caused a more sustained suppression of tumor growth compared with Camptosar treatment alone. These results indicate that SSA has potential safety and efficacy advantages for colon cancer chemoprevention as well as utility for treating malignant disease if combined with chemotherapy.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures

References
-
- Thun MJ, Henley SJ, Patrono C. Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. J Natl Cancer Inst. 2002;94:252–256. - PubMed
-
- Chan TA. Nonsteroidal anti-inflammatory drugs, apoptosis, and colon-cancer chemoprevention. Lancet Oncol. 2002;3:166–174. - PubMed
-
- Giardiello FM, Hamilton SR, Krush AJ. Treatment of colonic and rectal adenomas with sulindac in familial adenomatous polyposis. N Engl J Med. 1993;328:1313–1316. - PubMed
-
- Nugent KP, Farmer KC, Spigelman AD, Williams CB, Phillips RK. Randomized controlled trial of the effect of sulindac on duodenal and rectal polyposis and cell proliferation in patients with familial adenomatous polyposis. Br J Surg. 1993;80:1618–1619. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

