Efficacy, safety, and biomarkers of neoadjuvant bevacizumab, radiation therapy, and fluorouracil in rectal cancer: a multidisciplinary phase II study
- PMID: 19470921
- PMCID: PMC2702234
- DOI: 10.1200/JCO.2008.21.1771
Efficacy, safety, and biomarkers of neoadjuvant bevacizumab, radiation therapy, and fluorouracil in rectal cancer: a multidisciplinary phase II study
Abstract
Purpose: To assess the safety and efficacy of neoadjuvant bevacizumab with standard chemoradiotherapy in locally advanced rectal cancer and explore biomarkers for response.
Patients and methods: In a phase I/II study, 32 patients received four cycles of therapy consisting of: bevacizumab infusion (5 or 10 mg/kg) on day 1 of each cycle; fluorouracil infusion (225 mg/m(2)/24 hours) during cycles 2 to 4; external-beam irradiation (50.4 Gy in 28 fractions over 5.5 weeks); and surgery 7 to 10 weeks after completion of all therapies. We measured molecular, cellular, and physiologic biomarkers before treatment, during bevacizumab monotherapy, and during and after combination therapy.
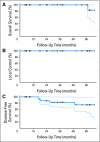
Results: Tumors regressed from a mass with mean size of 5 cm (range, 3 to 12 cm) to an ulcer/scar with mean size of 2.4 cm (range, 0.7 to 6.0 cm) in all 32 patients. Histologic examination revealed either no cancer or varying numbers of scattered cancer cells in a bed of fibrosis at the primary site. This treatment resulted in an actuarial 5-year local control and overall survival of 100%. Actuarial 5-year disease-free survival was 75% and five patients developed metastases postsurgery. Bevacizumab with chemoradiotherapy showed acceptable toxicity. Bevacizumab decreased tumor interstitial fluid pressure and blood flow. Baseline plasma soluble vascular endothelial growth factor receptor 1 (sVEGFR1), plasma vascular endothelial growth factor (VEGF), placental-derived growth factor (PlGF), and interleukin 6 (IL-6) during treatment, and circulating endothelial cells (CECs) after treatment showed significant correlations with outcome.
Conclusion: Bevacizumab with chemoradiotherapy appears safe and active and yields promising survival results in locally advanced rectal cancer. Plasma VEGF, PlGF, sVEGFR1, and IL-6 and CECs should be further evaluated as candidate biomarkers of response for this regimen.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures


References
-
- Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–2342. - PubMed
-
- Willett CG, Boucher Y, Duda DG, et al. Surrogate markers for antiangiogenic therapy and dose-limiting toxicities for bevacizumab with radiation and chemotherapy: Continued experience of a phase I trial in rectal cancer patients. J Clin Oncol. 2005;23:8136–8139. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

