Type 17 CD8+ T cells display enhanced antitumor immunity
- PMID: 19471017
- PMCID: PMC2713473
- DOI: 10.1182/blood-2009-02-203935
Type 17 CD8+ T cells display enhanced antitumor immunity
Abstract
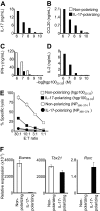
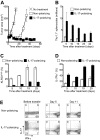
Interleukin-17 (IL-17)-secreting CD8(+) T cells have been described, but they have not been thoroughly studied and they do not have a known role in cancer immunotherapy. We skewed CD8(+) T cells to secrete IL-17 through priming in Th17-polarizing conditions. IL-17-producing CD8(+) T cells demonstrated reduced expression of Eomes and diminished cytolytic differentiation in vitro. However, after adoptive transfer, these cells converted to interferon-gamma-producing effector cells and mediated regression of large, established tumors. This improved antitumor immunity was associated with increased expression of IL-7R-alpha, decreased expression of killer cell lectin-like receptor G1, and enhanced persistence of the transferred cells. This report is the first description of a cancer therapy with IL-17-secreting CD8(+) T cells. These findings have implications for the improvement of CD8(+) T cell-based adoptive immunotherapy.
Figures
Similar articles
-
Tumor-specific Tc1, but not Tc2, cells deliver protective antitumor immunity.J Immunol. 2001 Dec 1;167(11):6497-502. doi: 10.4049/jimmunol.167.11.6497. J Immunol. 2001. PMID: 11714817
-
Reinforcement of cancer immunotherapy by adoptive transfer of cblb-deficient CD8+ T cells combined with a DC vaccine.Immunol Cell Biol. 2012 Jan;90(1):130-4. doi: 10.1038/icb.2011.11. Epub 2011 Mar 8. Immunol Cell Biol. 2012. PMID: 21383769
-
Tc1 and Tc2 effector cell therapy elicit long-term tumor immunity by contrasting mechanisms that result in complementary endogenous type 1 antitumor responses.J Immunol. 2004 Feb 1;172(3):1380-90. doi: 10.4049/jimmunol.172.3.1380. J Immunol. 2004. PMID: 14734713
-
IL-2 and IL-21 confer opposing differentiation programs to CD8+ T cells for adoptive immunotherapy.Blood. 2008 Jun 1;111(11):5326-33. doi: 10.1182/blood-2007-09-113050. Epub 2008 Feb 14. Blood. 2008. PMID: 18276844 Free PMC article.
-
Harnessing the CD8+ T-cell subsets with stemness for tumor immunotherapy.Future Oncol. 2018 Oct;14(24):2433-2436. doi: 10.2217/fon-2018-0238. Epub 2018 Aug 24. Future Oncol. 2018. PMID: 30141349 Review. No abstract available.
Cited by
-
Role of Dendritic Epidermal T Cells in Cutaneous Carcinoma.Front Immunol. 2020 Jul 14;11:1266. doi: 10.3389/fimmu.2020.01266. eCollection 2020. Front Immunol. 2020. PMID: 32765487 Free PMC article. Review.
-
Dendritic Cells in Irradiated Mice Trigger the Functional Plasticity and Antitumor Activity of Adoptively Transferred Tc17 Cells via IL12 Signaling.Clin Cancer Res. 2015 Jun 1;21(11):2546-57. doi: 10.1158/1078-0432.CCR-14-2294. Epub 2015 Apr 22. Clin Cancer Res. 2015. PMID: 25904754 Free PMC article.
-
IL-17A is produced by breast cancer TILs and promotes chemoresistance and proliferation through ERK1/2.Sci Rep. 2013 Dec 9;3:3456. doi: 10.1038/srep03456. Sci Rep. 2013. PMID: 24316750 Free PMC article.
-
Anticancer Tc9 cells: Long-lived tumor-killing T cells for adoptive therapy.Oncoimmunology. 2014 Apr 29;3:e28542. doi: 10.4161/onci.28542. eCollection 2014. Oncoimmunology. 2014. PMID: 25054087 Free PMC article.
-
Friend or Foe - Tc17 cell generation and current evidence for their importance in human disease.Discov Immunol. 2023 Jul 20;2(1):kyad010. doi: 10.1093/discim/kyad010. eCollection 2023. Discov Immunol. 2023. PMID: 38567057 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

