Optical imaging of postsynaptic odor representation in the glomerular layer of the mouse olfactory bulb
- PMID: 19474178
- PMCID: PMC2724327
- DOI: 10.1152/jn.00020.2009
Optical imaging of postsynaptic odor representation in the glomerular layer of the mouse olfactory bulb
Abstract
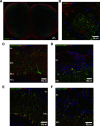
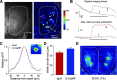
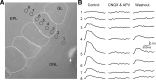
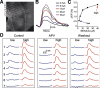
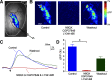
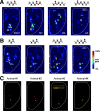
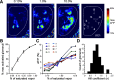
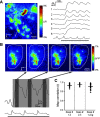
Olfactory glomeruli are the loci where the first odor-representation map emerges. The glomerular layer comprises exquisite local synaptic circuits for the processing of olfactory coding patterns immediately after their emergence. To understand how an odor map is transferred from afferent terminals to postsynaptic dendrites, it is essential to directly monitor the odor-evoked glomerular postsynaptic activity patterns. Here we report the use of a transgenic mouse expressing a Ca(2+)-sensitive green fluorescence protein (GCaMP2) under a Kv3.1 potassium-channel promoter. Immunostaining revealed that GCaMP2 was specifically expressed in mitral and tufted cells and a subpopulation of juxtaglomerular cells but not in olfactory nerve terminals. Both in vitro and in vivo imaging combined with glutamate receptor pharmacology confirmed that odor maps reported by GCaMP2 were of a postsynaptic origin. These mice thus provided an unprecedented opportunity to analyze the spatial activity pattern reflecting purely postsynaptic olfactory codes. The odor-evoked GCaMP2 signal had both focal and diffuse spatial components. The focalized hot spots corresponded to individually activated glomeruli. In GCaMP2-reported postsynaptic odor maps, different odorants activated distinct but overlapping sets of glomeruli. Increasing odor concentration increased both individual glomerular response amplitude and the total number of activated glomeruli. Furthermore, the GCaMP2 response displayed a fast time course that enabled us to analyze the temporal dynamics of odor maps over consecutive sniff cycles. In summary, with cell-specific targeting of a genetically encoded Ca(2+) indicator, we have successfully isolated and characterized an intermediate level of odor representation between olfactory nerve input and principal mitral/tufted cell output.
Figures
References
-
- Abraham NM, Spors H, Carleton A, Margrie TW, Kuner T, Schaefer AT. Maintaining accuracy at the expense of speed: stimulus similarity defines odor discrimination time in mice. Neuron 44: 865–876, 2004. - PubMed
-
- Aroniadou-Anderjaska V, Zhou FM, Priest CA, Ennis M, Shipley MT. Tonic and synaptically evoked presynaptic inhibition of sensory input to the rat olfactory bulb via GABA(B) heteroreceptors. J Neurophysiol 84: 1194–1203, 2000. - PubMed
-
- Aungst JL, Heyward PM, Puche AC, Karnup SV, Hayar A, Szabo G, Shipley MT. Centre-surround inhibition among olfactory bulb glomeruli. Nature 426: 623–629, 2003. - PubMed
-
- Berkowicz DA, Trombley PQ, Shepherd GM. Evidence for glutamate as the olfactory receptor cell neurotransmitter. J Neurophysiol 71: 2557–2561, 1994. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

