Genetic evidence for shared mechanisms of epimorphic regeneration in zebrafish
- PMID: 19474300
- PMCID: PMC2695073
- DOI: 10.1073/pnas.0811186106
Genetic evidence for shared mechanisms of epimorphic regeneration in zebrafish
Abstract
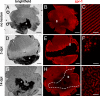
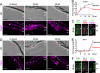
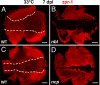
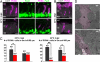
In a microarray-based gene profiling analysis of Müller glia-derived retinal stem cells in light-damaged retinas from adult zebrafish, we found that 2 genes required for regeneration of fin and heart tissues in zebrafish, hspd1 (heat shock 60-kDa protein 1) and mps1 (monopolar spindle 1), were up-regulated. Expression of both genes in the neurogenic Müller glia and progenitors was independently verified by quantitative reverse transcriptase PCR and in situ hybridization. Functional analysis of temperature-sensitive mutants of hspd1 and mps1 revealed that both are necessary for Müller glia-based cone photoreceptor regeneration in adult zebrafish retina. In the amputated fin, hspd1 is required for the induction of mesenchymal stem cells and blastema formation, whereas mps1 is required at a later step for rapid cell proliferation and outgrowth. This temporal sequence of hspd1 and mps1 function is conserved in the regenerating retina. Comparison of gene expression profiles from regenerating zebrafish retina, caudal fin, and heart muscle revealed additional candidate genes potentially implicated in injury-induced epimorphic regeneration in diverse zebrafish tissues.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Sánchez Alvarado A, Tsonis PA. Bridging the regeneration gap: Genetic insights from diverse animal models. Nat Rev Genet. 2006;7:873–884. - PubMed
-
- Poss KD, Wilson LG, Keating MT. Heart regeneration in zebrafish. Science. 2002;298:2188–2190. - PubMed
-
- Bernhardt RR, Tongiorgi E, Anzini P, Schachner M. Increased expression of specific recognition molecules by retinal ganglion cells and by optic pathway glia accompanies the successful regeneration of retinal axons in adult zebrafish. J Comp Neurol. 1996;376:253–264. - PubMed
-
- Becker T, Wullimann MF, Becker CG, Bernhardt RR, Schachner M. Axonal regrowth after spinal cord transection in adult zebrafish. J Comp Neurol. 1997;377:577–595. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

