A novel human gamma-globin gene vector for genetic correction of sickle cell anemia in a humanized sickle mouse model: critical determinants for successful correction
- PMID: 19474450
- PMCID: PMC2723013
- DOI: 10.1182/blood-2009-01-201863
A novel human gamma-globin gene vector for genetic correction of sickle cell anemia in a humanized sickle mouse model: critical determinants for successful correction
Abstract
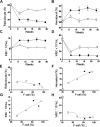
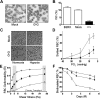
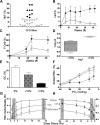
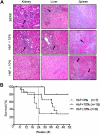
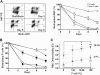
We show that lentiviral delivery of human gamma-globin gene under beta-globin regulatory control elements in hematopoietic stem cells (HSCs) results in sufficient postnatal fetal hemoglobin (HbF) expression to correct sickle cell anemia (SCA) in the Berkeley "humanized" sickle mouse. Upon de-escalating the amount of transduced HSCs in transplant recipients, using reduced-intensity conditioning and varying gene transfer efficiency and vector copy number, we assessed critical parameters needed for correction. A systematic quantification of functional and hematologic red blood cell (RBC) indices, organ pathology, and life span was used to determine the minimal amount of HbF, F cells, HbF/F-cell, and gene-modified HSCs required for correcting the sickle phenotype. We show that long-term amelioration of disease occurred (1) when HbF exceeded 10%, F cells constituted two-thirds of the circulating RBCs, and HbF/F cell was one-third of the total hemoglobin in sickle RBCs; and (2) when approximately 20% gene-modified HSCs repopulated the marrow. Moreover, we show a novel model using reduced-intensity conditioning to determine genetically corrected HSC threshold that corrects a hematopoietic disease. These studies provide a strong preclinical model for what it would take to genetically correct SCA and are a foundation for the use of this vector in a human clinical trial.
Figures

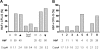
References
-
- Darbari DS, Kple-Faget P, Kwagyan J, Rana S, Gordeuk VR, Castro O. Circumstances of death in adult sickle cell disease patients. Am J Hematol. 2006;81(11):858–863. - PubMed
-
- Platt OS, Brambilla DJ, Rosse WF, et al. Mortality in sickle cell disease: life expectancy and risk factors for early death. N Engl J Med. 1994;330(23):1639–1644. - PubMed
-
- World Health Organization. Sickle-Cell Anaemia. Fifty-Ninth World Health Assembly, Provisional Agenda Item 11.4. [Accessed June 13, 2006];2006 A59:1. http://www.who.int/gb/ebwha/pdf_files/WHA59/A59_9-en.pdf.
-
- Walters MC, Patience M, Leisenring W, et al. Barriers to bone marrow transplantation for sickle cell anemia. Biol Blood Marrow Transplant. 1996;2(2):100–104. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

