Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis
- PMID: 19476657
- PMCID: PMC2700094
- DOI: 10.1186/1471-213X-9-31
Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis
Abstract
Background: In vertebrates, the inner ear is comprised of the cochlea and vestibular system, which develop from the otic vesicle. This process is regulated via inductive interactions from surrounding tissues. Tbx1, the gene responsible for velo-cardio-facial syndrome/DiGeorge syndrome in humans, is required for ear development in mice. Tbx1 is expressed in the otic epithelium and adjacent periotic mesenchyme (POM), and both of these domains are required for inner ear formation. To study the function of Tbx1 in the POM, we have conditionally inactivated Tbx1 in the mesoderm while keeping expression in the otic vesicle intact.
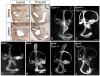
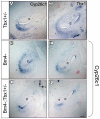
Results: Conditional mutants (TCre-KO) displayed malformed inner ears, including a hypoplastic otic vesicle and a severely shortened cochlear duct, indicating that Tbx1 expression in the POM is necessary for proper inner ear formation. Expression of the mesenchyme marker Brn4 was also lost in the TCre-KO. Brn4-;Tbx1+/-embryos displayed defects in growth of the distal cochlea. To identify a potential signal from the POM to the otic epithelium, expression of retinoic acid (RA) catabolizing genes was examined in both mutants. Cyp26a1 expression was altered in the TCre-KO, while Cyp26c1 showed reduced expression in both TCre-KO and Brn4-;Tbx1+/- embryos.
Conclusion: These results indicate that Tbx1 expression in the POM regulates cochlear outgrowth potentially via control of local retinoic acid activity.
Figures




Similar articles
-
22q11 deletion syndrome: a role for TBX1 in pharyngeal and cardiovascular development.Pediatr Cardiol. 2010 Apr;31(3):378-90. doi: 10.1007/s00246-009-9613-0. Pediatr Cardiol. 2010. PMID: 20054531 Review.
-
Cooperative function of Tbx1 and Brn4 in the periotic mesenchyme is necessary for cochlea formation.J Assoc Res Otolaryngol. 2008 Mar;9(1):33-43. doi: 10.1007/s10162-008-0110-6. Epub 2008 Jan 30. J Assoc Res Otolaryngol. 2008. PMID: 18231833 Free PMC article.
-
Identification of putative retinoic acid target genes downstream of mesenchymal Tbx1 during inner ear development.Dev Dyn. 2012 Mar;241(3):563-73. doi: 10.1002/dvdy.23731. Epub 2012 Feb 1. Dev Dyn. 2012. PMID: 22275070 Free PMC article.
-
TBX1 is required for inner ear morphogenesis.Hum Mol Genet. 2003 Aug 15;12(16):2041-8. doi: 10.1093/hmg/ddg216. Hum Mol Genet. 2003. PMID: 12913075
-
Retinoid signaling in inner ear development: A "Goldilocks" phenomenon.Am J Med Genet A. 2010 Dec;152A(12):2947-61. doi: 10.1002/ajmg.a.33670. Am J Med Genet A. 2010. PMID: 21108385 Free PMC article. Review.
Cited by
-
Dissecting the molecular basis of organ of Corti development: Where are we now?Hear Res. 2011 Jun;276(1-2):16-26. doi: 10.1016/j.heares.2011.01.007. Epub 2011 Jan 21. Hear Res. 2011. PMID: 21256948 Free PMC article. Review.
-
Chromatin remodeler CHD7 is critical for cochlear morphogenesis and neurosensory patterning.Dev Biol. 2021 Sep;477:11-21. doi: 10.1016/j.ydbio.2021.05.009. Epub 2021 May 15. Dev Biol. 2021. PMID: 34004180 Free PMC article.
-
22q11 deletion syndrome: a role for TBX1 in pharyngeal and cardiovascular development.Pediatr Cardiol. 2010 Apr;31(3):378-90. doi: 10.1007/s00246-009-9613-0. Pediatr Cardiol. 2010. PMID: 20054531 Review.
-
Dysphagia and disrupted cranial nerve development in a mouse model of DiGeorge (22q11) deletion syndrome.Dis Model Mech. 2014 Feb;7(2):245-57. doi: 10.1242/dmm.012484. Epub 2013 Dec 19. Dis Model Mech. 2014. PMID: 24357327 Free PMC article.
-
Immunodeficiency in DiGeorge Syndrome and Options for Treating Cases with Complete Athymia.Front Immunol. 2013 Oct 31;4:322. doi: 10.3389/fimmu.2013.00322. Front Immunol. 2013. PMID: 24198816 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

