Growth factor-dependent trafficking of cerebellar NMDA receptors via protein kinase B/Akt phosphorylation of NR2C
- PMID: 19477150
- PMCID: PMC2716006
- DOI: 10.1016/j.neuron.2009.04.015
Growth factor-dependent trafficking of cerebellar NMDA receptors via protein kinase B/Akt phosphorylation of NR2C
Abstract
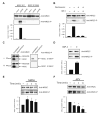
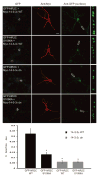
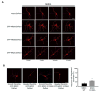
NMDA receptor subunit composition varies throughout the brain, providing molecular diversity in NMDA receptor function. The NR2 subunits (NR2A-D) in large part dictate the distinct functional properties of NMDA receptors and differentially regulate receptor trafficking. Although the NR2C subunit is highly enriched in cerebellar granule cells and plays a unique role in cerebellar function, little is known about NR2C-specific regulation of NMDA receptors. Here, we demonstrate that PKB/Akt directly phosphorylates NR2C on serine 1096 (S1096). In addition, we identify 14-3-3epsilon as an NR2C interactor, whose binding is dependent on S1096 phosphorylation. Both growth factor stimulation and NMDA receptor activity lead to a robust increase in both phosphorylation of NR2C on S1096 and surface expression of cerebellar NMDA receptors. Finally, we find that NR2C expression, unlike NR2A and NR2B, supports neuronal survival. Thus, our data provide a direct mechanistic link between growth factor stimulation and regulation of cerebellar NMDA receptors.
Figures


References
-
- Akazawa C, Shigemoto R, Bessho Y, Nakanishi S, Mizuno N. Differential expression of five N-methyl-D-aspartate receptor subunit mRNAs in the cerebellum of developing and adult rats. J Comp Neurol. 1994;347:150–160. - PubMed
-
- Bellone C, Nicoll RA. Rapid bidirectional switching of synaptic NMDA receptors. Neuron. 2007;55:779–785. - PubMed
-
- Berg D, Holzmann C, Riess O. 14-3-3 proteins in the nervous system. Nat Rev Neurosci. 2003;4:752–762. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

