Unconventional myosin traffic in cells reveals a selective actin cytoskeleton
- PMID: 19478066
- PMCID: PMC2701044
- DOI: 10.1073/pnas.0810451106
Unconventional myosin traffic in cells reveals a selective actin cytoskeleton
Abstract
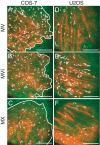
Eukaryotic cells have a self-organizing cytoskeleton where motors transport cargoes along cytoskeletal tracks. To understand the sorting process, we developed a system to observe single-molecule motility in a cellular context. We followed myosin classes V, VI, and X on triton-extracted actin cytoskeletons from Drosophila S2, mammalian COS-7, and mammalian U2OS cells. We find that these cells vary considerably in their global traffic patterns. The S2 and U2OS cells have regions of actin that either enhance or inhibit specific myosin classes. U2OS cells allow for 1 motor class, myosin VI, to move along stress fiber bundles, while motility of myosin V and X are suppressed. Myosin X motors are recruited to filopodia and the lamellar edge in S2 cells, whereas myosin VI motility is excluded from the same regions. Furthermore, we also see different velocities of myosin V motors in central regions of S2 cells, suggesting regional control of motor motility by the actin cytoskeleton. We also find unexpected features of the actin cytoskeletal network, including a population of reversed filaments with the barbed-end toward the cell center. This myosin motor regulation demonstrates that native actin cytoskeletons are more than just a collection of filaments.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
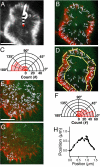
 is defined as the orientation angle and measured from the centroid of the cell to the beginning of a motor run (arrow). (Scale bar, 10 μm.) (B) Trajectory map (white arrows) of all myosin V motor (green) runs on the preserved actin cytoskeleton (red). Each arrow represents a single moving motor run. A processive run is defined as a linear movement lasting at least 3 consecutive frames (≥ 1.5 s). Each frame is acquired with a 0.5-s exposure for 200 frames. The majority of motors (76%) move toward the cell periphery. Very few MV motors (n = 7/138) actually switch actin tracks throughout this analysis (cyan). (C) Orientation angle measurements for processive runs. The majority of MV motors have a trajectory angle of 100–180°, indicating that the motors move toward the cell periphery. (D) The zone of motility (yellow outline) is where most motors move toward the cell periphery with a similar velocity. This motility zone is directly adjacent to a central zone of slow motility. (E) Trajectory map (white arrows) of single myosin VI motors (green) moving on a preserved actin cytoskeleton (red). Each arrow represents a single motor run. The majority of motors (72%) move away from the cell periphery. (Scale bar, 10 μm.) (F) Orientation angles measured for all MVI motors making a processive run. Most motors have a trajectory angle of 0–80°, indicating that these motors move away from the cell periphery or toward the cell center. (G) MVI motors that switch actin tracks are depicted (white). There are more MVI motors that switch than MV motors (compare to panel B, cyan). (H) Tracking pattern of a MVI motor that switches actin tracks (boxed in G). The motor runs for 26 frames over a distance of 1.5 μm. Since this motor tracks with a sharp turn (>20°), it is characterized as a motor that switches actin tracks during motility.
is defined as the orientation angle and measured from the centroid of the cell to the beginning of a motor run (arrow). (Scale bar, 10 μm.) (B) Trajectory map (white arrows) of all myosin V motor (green) runs on the preserved actin cytoskeleton (red). Each arrow represents a single moving motor run. A processive run is defined as a linear movement lasting at least 3 consecutive frames (≥ 1.5 s). Each frame is acquired with a 0.5-s exposure for 200 frames. The majority of motors (76%) move toward the cell periphery. Very few MV motors (n = 7/138) actually switch actin tracks throughout this analysis (cyan). (C) Orientation angle measurements for processive runs. The majority of MV motors have a trajectory angle of 100–180°, indicating that the motors move toward the cell periphery. (D) The zone of motility (yellow outline) is where most motors move toward the cell periphery with a similar velocity. This motility zone is directly adjacent to a central zone of slow motility. (E) Trajectory map (white arrows) of single myosin VI motors (green) moving on a preserved actin cytoskeleton (red). Each arrow represents a single motor run. The majority of motors (72%) move away from the cell periphery. (Scale bar, 10 μm.) (F) Orientation angles measured for all MVI motors making a processive run. Most motors have a trajectory angle of 0–80°, indicating that these motors move away from the cell periphery or toward the cell center. (G) MVI motors that switch actin tracks are depicted (white). There are more MVI motors that switch than MV motors (compare to panel B, cyan). (H) Tracking pattern of a MVI motor that switches actin tracks (boxed in G). The motor runs for 26 frames over a distance of 1.5 μm. Since this motor tracks with a sharp turn (>20°), it is characterized as a motor that switches actin tracks during motility.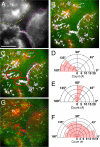

Similar articles
-
Actin age orchestrates myosin-5 and myosin-6 run lengths.Curr Biol. 2015 Aug 3;25(15):2057-62. doi: 10.1016/j.cub.2015.06.033. Epub 2015 Jul 16. Curr Biol. 2015. PMID: 26190073 Free PMC article.
-
Cargo recognition and cargo-mediated regulation of unconventional myosins.Acc Chem Res. 2014 Oct 21;47(10):3061-70. doi: 10.1021/ar500216z. Epub 2014 Sep 17. Acc Chem Res. 2014. PMID: 25230296 Review.
-
The stepping pattern of myosin X is adapted for processive motility on bundled actin.Biophys J. 2010 Sep 22;99(6):1818-26. doi: 10.1016/j.bpj.2010.06.066. Biophys J. 2010. PMID: 20858426 Free PMC article.
-
A myosin motor that selects bundled actin for motility.Proc Natl Acad Sci U S A. 2008 Jul 15;105(28):9616-20. doi: 10.1073/pnas.0802592105. Epub 2008 Jul 3. Proc Natl Acad Sci U S A. 2008. PMID: 18599451 Free PMC article.
-
Structural Basis of Cargo Recognition by Unconventional Myosins in Cellular Trafficking.Traffic. 2016 Aug;17(8):822-38. doi: 10.1111/tra.12383. Epub 2016 Mar 17. Traffic. 2016. PMID: 26842936 Review.
Cited by
-
Fascin- and α-Actinin-Bundled Networks Contain Intrinsic Structural Features that Drive Protein Sorting.Curr Biol. 2016 Oct 24;26(20):2697-2706. doi: 10.1016/j.cub.2016.07.080. Epub 2016 Sep 22. Curr Biol. 2016. PMID: 27666967 Free PMC article.
-
Bridging membrane and cytoskeleton dynamics in the secretory and endocytic pathways.Nat Cell Biol. 2011 Dec 22;14(1):11-9. doi: 10.1038/ncb2409. Nat Cell Biol. 2011. PMID: 22193159 Review.
-
Human myosin VIIa is a very slow processive motor protein on various cellular actin structures.J Biol Chem. 2017 Jun 30;292(26):10950-10960. doi: 10.1074/jbc.M116.765966. Epub 2017 May 15. J Biol Chem. 2017. PMID: 28507101 Free PMC article.
-
Activated full-length myosin-X moves processively on filopodia with large steps toward diverse two-dimensional directions.Sci Rep. 2017 Mar 13;7:44237. doi: 10.1038/srep44237. Sci Rep. 2017. PMID: 28287133 Free PMC article.
-
The 133-kDa N-terminal domain enables myosin 15 to maintain mechanotransducing stereocilia and is essential for hearing.Elife. 2015 Aug 24;4:e08627. doi: 10.7554/eLife.08627. Elife. 2015. PMID: 26302205 Free PMC article.
References
-
- Bahler M. Are class III and class IX myosins motorized signalling molecules? Biochim Biophys Acta. 2000;1496:52–59. - PubMed
-
- Sellers J. Myosins. Oxford: Oxford University Press; 1999.
-
- Tuxworth RI, Titus MA. Unconventional myosins: Anchors in the membrane traffic relay. Traffic. 2000;1:11–18. - PubMed
-
- Tang N, Ostap EM. Motor domain-dependent localization of myo1b (myr-1) Curr Biol. 2001;11:1131–1135. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

