Settings and mechanisms for trans-cellular diapedesis
- PMID: 19482605
- PMCID: PMC3177403
- DOI: 10.2741/3587
Settings and mechanisms for trans-cellular diapedesis
Abstract
Immune system functions require blood leukocytes to continuously traffic throughout the body and repeatedly cross endothelial barriers (i.e., diapedese) as they enter (intravasation) and exit (extravasation) the circulation. The earliest studies to directly characterize diapedesis in vivo suggested co-existence of two distinct migratory pathways: between (para-cellular) and through (trans-cellular) individual endothelial cells. However, in the absence of conclusive in vitro observations, the latter pathway remained poorly accepted. The recent emergence of unambiguous in vitro reports of trans-cellular diapedesis has begun to illuminate mechanisms for this pathway and has renewed interest in its physiological roles. A thorough reevaluation of the existing literature reveals a large number of studies documenting significant use of the trans-cellular pathway in diverse in vivo settings. These include constitutive trafficking in bone marrow and lymphoid organs as well as upregulated extravasation in peripheral tissues during inflammation. Here we collectively summarize these in vivo observations alongside the emerging in vitro data in order to provide a framework for understanding the settings, mechanisms and roles for the trans-cellular route of diapedesis.
Figures
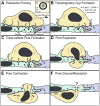
Similar articles
-
Mechanisms for transcellular diapedesis: probing and pathfinding by 'invadosome-like protrusions'.J Cell Sci. 2009 Sep 1;122(Pt 17):3025-35. doi: 10.1242/jcs.047522. J Cell Sci. 2009. PMID: 19692589 Review.
-
Trans-cellular migration: cell-cell contacts get intimate.Curr Opin Cell Biol. 2008 Oct;20(5):533-40. doi: 10.1016/j.ceb.2008.05.007. Epub 2008 Jul 1. Curr Opin Cell Biol. 2008. PMID: 18595683 Free PMC article. Review.
-
Review: leucocyte-endothelial cell crosstalk at the blood-brain barrier: a prerequisite for successful immune cell entry to the brain.Neuropathol Appl Neurobiol. 2011 Feb;37(1):24-39. doi: 10.1111/j.1365-2990.2010.01140.x. Neuropathol Appl Neurobiol. 2011. PMID: 20946472 Review.
-
Molecular events during leukocyte diapedesis.FEBS J. 2006 Oct;273(19):4399-407. doi: 10.1111/j.1742-4658.2006.05439.x. Epub 2006 Sep 11. FEBS J. 2006. PMID: 16965541 Review.
-
Getting leukocytes to the site of inflammation.Vet Pathol. 2013 Jan;50(1):7-22. doi: 10.1177/0300985812469883. Vet Pathol. 2013. PMID: 23345459 Free PMC article. Review.
Cited by
-
ALCAM (CD166) is involved in extravasation of monocytes rather than T cells across the blood-brain barrier.J Cereb Blood Flow Metab. 2017 Aug;37(8):2894-2909. doi: 10.1177/0271678X16678639. Epub 2016 Nov 14. J Cereb Blood Flow Metab. 2017. PMID: 28273717 Free PMC article.
-
Transendothelial migration: unifying principles from the endothelial perspective.Immunol Rev. 2016 Sep;273(1):61-75. doi: 10.1111/imr.12443. Immunol Rev. 2016. PMID: 27558328 Free PMC article. Review.
-
Rho-GTPase signaling in leukocyte extravasation: an endothelial point of view.Cell Adh Migr. 2014;8(2):67-75. doi: 10.4161/cam.28244. Cell Adh Migr. 2014. PMID: 24621576 Free PMC article. Review.
-
Endothelial transmigration by Trypanosoma cruzi.PLoS One. 2013 Dec 2;8(12):e81187. doi: 10.1371/journal.pone.0081187. eCollection 2013. PLoS One. 2013. PMID: 24312535 Free PMC article. Clinical Trial.
-
Endocanalicular transendothelial crossing (ETC): A novel intravasation mode used by HEK-EBNA293-VEGF-D cells during the metastatic process in a xenograft model.PLoS One. 2020 Oct 21;15(10):e0239932. doi: 10.1371/journal.pone.0239932. eCollection 2020. PLoS One. 2020. PMID: 33085676 Free PMC article.
References
-
- von Andrian UH, Mackay CR. T-cell function and migration. Two sides of the same coin. N Engl J Med. 2000;343:1020–1034. - PubMed
-
- Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol. 2007;7:678–89. - PubMed
-
- Springer TA. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994;76:301–14. - PubMed
-
- Luscinskas FW, Kansas GS, Ding H, Pizcueta P, Schleiffenbaum BE, Tedder TF, Gimbrone MA., Jr Monocyte rolling, arrest and spreading on IL-4-activated vascular endothelium under flow is mediated via sequential action of L-selectin, beta 1-integrins, and beta 2-integrins. J Cell Biol. 1994;125:1417–27. - PMC - PubMed
-
- Carman CV, Springer TA. Integrin avidity regulation: are changes in affinity and conformation underemphasized? Curr Opin Cell Biol. 2003;15:547–56. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

