The role of phosphodiesterase 3 in endotoxin-induced acute kidney injury
- PMID: 19486524
- PMCID: PMC2694814
- DOI: 10.1186/1471-2334-9-80
The role of phosphodiesterase 3 in endotoxin-induced acute kidney injury
Abstract
Background: Acute kidney injury frequently accompanies sepsis. Endotoxin is known to reduce tissue levels of cAMP and low levels of cAMP have been associated with renal injury. We, therefore, hypothesized that endotoxin induced renal injury by activating phosphodiesterase 3 (PDE3) which metabolizes cAMP and that amrinone an inhibitor of PDE3 would prevent the renal injury.
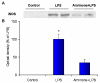
Methods: Animals were divided into three groups (n = 7/group): 1) Control (0.9% NaCl infusion without LPS); 2) LPS (0.9% NaCl infusion with LPS); 3) Amrinone+LPS (Amrinone infusion with LPS). Either lipopolysaccharide (LPS) or vehicle was injected via the jugular vein and the rats followed for 3 hours. We explored the expression of PDE3 isoenzymes and the concentrations of cAMP in the tissue.
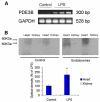
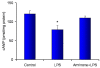
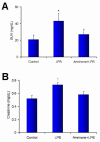
Results: The PDE3B gene but not PDE3A was upregulated in the kidney of LPS group. Immunohistochemistry also showed that PDE3B was expressed in the distal tubule in the controls and LPS caused PDE3B expression in the proximal as well. However, PDE3A was not expressed in the kidney either in the control or LPS treated groups. Tissue level of cAMP was decreased after LPS and was associated with an increase in blood urea nitrogen, creatinine, ultrastructural proximal tubular changes, and expression of inducible nitric oxide synthase (iNOS) in the endotoxemic kidney. In septic animals the phosphodiesterase 3 inhibitor, amrinone, preserved the tissue cAMP level, renal structural changes, and attenuated the increased blood urea nitrogen, creatinine, and iNOS expression in the kidney.
Conclusion: These findings suggest a significant role for PDE3B as an important mediator of LPS-induced acute kidney injury.
Figures


Similar articles
-
Yohimbine ameliorates lipopolysaccharide-induced acute kidney injury in rats.Eur J Pharmacol. 2020 Mar 15;871:172917. doi: 10.1016/j.ejphar.2020.172917. Epub 2020 Jan 11. Eur J Pharmacol. 2020. PMID: 31935395
-
Inhibition of inducible nitric oxide synthase reduces lipopolysaccharide-induced renal injury in the rat.Clin Exp Pharmacol Physiol. 2004 Dec;31(12):842-6. doi: 10.1111/j.1440-1681.2004.04096.x. Clin Exp Pharmacol Physiol. 2004. PMID: 15659046
-
Lipopolysaccharide-induced acute renal failure in conscious rats: effects of specific phosphodiesterase type 3 and 4 inhibition.J Pharmacol Exp Ther. 2002 Oct;303(1):364-74. doi: 10.1124/jpet.102.036194. J Pharmacol Exp Ther. 2002. PMID: 12235272
-
Role of phosphodiesterase type 3A and 3B in regulating platelet and cardiac function using subtype-selective knockout mice.Cell Signal. 2007 Aug;19(8):1765-71. doi: 10.1016/j.cellsig.2007.03.012. Epub 2007 Apr 6. Cell Signal. 2007. PMID: 17482796
-
Nitric oxide differentially regulates renal ATP-binding cassette transporters during endotoxemia.Pflugers Arch. 2007 May;454(2):321-34. doi: 10.1007/s00424-007-0210-x. Epub 2007 Feb 7. Pflugers Arch. 2007. PMID: 17285300 Free PMC article.
Cited by
-
The renal vasodilatory effect of prostaglandins is ameliorated in isolated-perfused kidneys of endotoxemic mice.Pflugers Arch. 2018 Nov;470(11):1691-1703. doi: 10.1007/s00424-018-2183-3. Epub 2018 Jul 19. Pflugers Arch. 2018. PMID: 30027346
-
Use of inotropes and vasopressor agents in critically ill patients.Br J Pharmacol. 2012 Apr;165(7):2015-33. doi: 10.1111/j.1476-5381.2011.01588.x. Br J Pharmacol. 2012. PMID: 21740415 Free PMC article. Review.
References
-
- Lee JH, Del Sorbo L, Uhlig S, Porro GA, Whitehead T, Voglis S, Liu M, Slutsky AS, Zhang H. Intercellular adhesion molecule-1 mediates cellular cross-talk between parenchymal and immune cells after lipopolysaccharide neutralization. J Immunol. 2004;172(1):608–616. - PubMed
-
- Ogawa S, Koga S, Kuwabara K, Brett J, Morrow B, Morris SA, Bilezikian JP, Silverstein SC, Stern D. Hypoxia-induced increased permeability of endothelial monolayers occurs through lowering of cellular cAMP levels. Am J Physiol. 1992;262(3 Pt 1):C546–554. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

