Complement promotes the development of inflammatory T-helper 17 cells through synergistic interaction with Toll-like receptor signaling and interleukin-6 production
- PMID: 19491392
- PMCID: PMC2721782
- DOI: 10.1182/blood-2009-01-198283
Complement promotes the development of inflammatory T-helper 17 cells through synergistic interaction with Toll-like receptor signaling and interleukin-6 production
Abstract
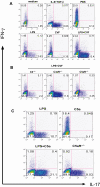
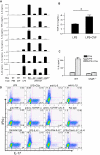
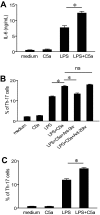
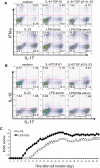
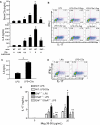
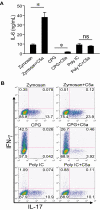
Toll-like receptors (TLRs) and complement are 2 major components of innate immunity that provide a first-line host defense and shape the adaptive immune responses. We show here that coincidental activation of complement and several TLRs in mice led to the synergistic production of serum factors that promoted T-helper cell 17 (Th17) differentiation from anti-CD3/CD28 or antigen-stimulated T cells. Although multiple TLR-triggered cytokines were regulated by complement, Th17 cell-promoting activity in the serum was correlated with interleukin (IL)-6 induction, and antibody neutralization of IL-6 abrogated the complement effect. By using both in vitro and in vivo approaches, we examined in more detail the mechanism and physiologic implication of complement/TLR4 interaction on Th17-cell differentiation. We found that the complement effect required C5a receptor, was evident at physiologically relevant levels of C5a, and could be demonstrated in cultured peritoneal macrophages as well as in the setting of antigen immunization. Importantly, despite an inhibitory effect of complement on IL-23 production, complement-promoted Th17 cells were functionally competent in causing autoimmunity in an adoptive transfer model of experimental autoimmune encephalomyelitis. Collectively, these data establish a link between complement/TLR interaction and Th17-cell differentiation and provide new insight into the mechanism of action of complement in autoimmunity.
Figures
Similar articles
-
Complement activation product C5a is a selective suppressor of TLR4-induced, but not TLR3-induced, production of IL-27(p28) from macrophages.J Immunol. 2012 May 15;188(10):5086-93. doi: 10.4049/jimmunol.1102914. Epub 2012 Apr 9. J Immunol. 2012. PMID: 22491257 Free PMC article.
-
Complement drives Th17 cell differentiation and triggers autoimmune arthritis.J Exp Med. 2010 Jun 7;207(6):1135-43. doi: 10.1084/jem.20092301. Epub 2010 May 10. J Exp Med. 2010. PMID: 20457757 Free PMC article.
-
Toll-like receptor 2 signaling in CD4(+) T lymphocytes promotes T helper 17 responses and regulates the pathogenesis of autoimmune disease.Immunity. 2010 May 28;32(5):692-702. doi: 10.1016/j.immuni.2010.04.010. Epub 2010 Apr 29. Immunity. 2010. PMID: 20434372 Free PMC article.
-
Crosstalk between complement and toll-like receptors.Toxicol Pathol. 2012;40(2):174-82. doi: 10.1177/0192623311428478. Epub 2011 Nov 22. Toxicol Pathol. 2012. PMID: 22109714 Review.
-
Therapeutic benefits of regulating inflammation in autoimmunity.Inflamm Allergy Drug Targets. 2008 Sep;7(3):203-10. doi: 10.2174/187152808785748155. Inflamm Allergy Drug Targets. 2008. PMID: 18782028 Review.
Cited by
-
Complement Is Required for Microbe-Driven Induction of Th17 and Periodontitis.J Immunol. 2022 Oct 1;209(7):1370-1378. doi: 10.4049/jimmunol.2200338. Epub 2022 Aug 26. J Immunol. 2022. PMID: 36028293 Free PMC article.
-
Reduced graft-versus-host disease in C3-deficient mice is associated with decreased donor Th1/Th17 differentiation.Biol Blood Marrow Transplant. 2012 Aug;18(8):1174-81. doi: 10.1016/j.bbmt.2012.05.014. Epub 2012 Jun 1. Biol Blood Marrow Transplant. 2012. PMID: 22664751 Free PMC article.
-
α2,6-Sialylation is Upregulated in Severe COVID-19 Implicating the Complement Cascade.medRxiv [Preprint]. 2022 Jun 8:2022.06.06.22275981. doi: 10.1101/2022.06.06.22275981. medRxiv. 2022. Update in: ACS Infect Dis. 2022 Nov 11;8(11):2348-2361. doi: 10.1021/acsinfecdis.2c00421. PMID: 35702159 Free PMC article. Updated. Preprint.
-
The role of complement in the diagnosis and management of allergic rhinitis and allergic asthma.Curr Allergy Asthma Rep. 2011 Apr;11(2):122-30. doi: 10.1007/s11882-010-0171-6. Curr Allergy Asthma Rep. 2011. PMID: 21170614 Review.
-
Increased systemic Th17 cytokines are associated with diastolic dysfunction in children and adolescents with diabetic ketoacidosis.PLoS One. 2013 Aug 27;8(8):e71905. doi: 10.1371/journal.pone.0071905. eCollection 2013. PLoS One. 2013. PMID: 24013901 Free PMC article.
References
-
- Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4:499–511. - PubMed
-
- Iwasaki A, Medzhitov R. Toll-like receptor control of the adaptive immune responses. Nat Immunol. 2004;5:987–995. - PubMed
-
- Walport MJ. Complement. First of two parts. N Engl J Med. 2001;344:1058–1066. - PubMed
-
- Walport MJ. Complement. Second of two parts. N Engl J Med. 2001;344:1140–1144. - PubMed
-
- Poltorak A, He X, Smirnova I, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998;282:2085–2088. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

