Conformational targeting of fibrillar polyglutamine proteins in live cells escalates aggregation and cytotoxicity
- PMID: 19492089
- PMCID: PMC2683928
- DOI: 10.1371/journal.pone.0005727
Conformational targeting of fibrillar polyglutamine proteins in live cells escalates aggregation and cytotoxicity
Abstract
Background: Misfolding- and aggregation-prone proteins underlying Parkinson's, Huntington's and Machado-Joseph diseases, namely alpha-synuclein, huntingtin, and ataxin-3 respectively, adopt numerous intracellular conformations during pathogenesis, including globular intermediates and insoluble amyloid-like fibrils. Such conformational diversity has complicated research into amyloid-associated intracellular dysfunction and neurodegeneration. To this end, recombinant single-chain Fv antibodies (scFvs) are compelling molecular tools that can be selected against specific protein conformations, and expressed inside cells as intrabodies, for investigative and therapeutic purposes.
Methodology/principal findings: Using atomic force microscopy (AFM) and live-cell fluorescence microscopy, we report that a human scFv selected against the fibrillar form of alpha-synuclein targets isomorphic conformations of misfolded polyglutamine proteins. When expressed in the cytoplasm of striatal cells, this conformation-specific intrabody co-localizes with intracellular aggregates of misfolded ataxin-3 and a pathological fragment of huntingtin, and enhances the aggregation propensity of both disease-linked polyglutamine proteins. Using this intrabody as a tool for modulating the kinetics of amyloidogenesis, we show that escalating aggregate formation of a pathologic huntingtin fragment is not cytoprotective in striatal cells, but rather heightens oxidative stress and cell death as detected by flow cytometry. Instead, cellular protection is achieved by suppressing aggregation using a previously described intrabody that binds to the amyloidogenic N-terminus of huntingtin. Analogous cytotoxic results are observed following conformational targeting of normal or polyglutamine-expanded human ataxin-3, which partially aggregate through non-polyglutamine domains.
Conclusions/significance: These findings validate that the rate of aggregation modulates polyglutamine-mediated intracellular dysfunction, and caution that molecules designed to specifically hasten aggregation may be detrimental as therapies for polyglutamine disorders. Moreover, our findings introduce a novel antibody-based tool that, as a consequence of its general specificity for fibrillar conformations and its ability to function intracellularly, offers broad research potential for a variety of human amyloid diseases.
Conflict of interest statement
Figures
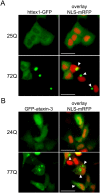
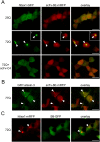
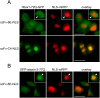
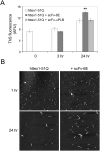
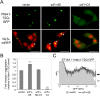
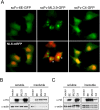
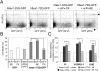
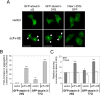
Similar articles
-
Amyloid formation by mutant huntingtin: threshold, progressivity and recruitment of normal polyglutamine proteins.Somat Cell Mol Genet. 1998 Jul;24(4):217-33. doi: 10.1023/b:scam.0000007124.19463.e5. Somat Cell Mol Genet. 1998. PMID: 10410676
-
Human single-chain Fv intrabodies counteract in situ huntingtin aggregation in cellular models of Huntington's disease.Proc Natl Acad Sci U S A. 2001 Apr 10;98(8):4764-9. doi: 10.1073/pnas.071058398. Proc Natl Acad Sci U S A. 2001. PMID: 11296304 Free PMC article.
-
Aging causes distinct characteristics of polyglutamine amyloids in vivo.Genes Cells. 2011 May;16(5):557-64. doi: 10.1111/j.1365-2443.2011.01505.x. Epub 2011 Apr 5. Genes Cells. 2011. PMID: 21466635
-
Polyglutamine diseases: the special case of ataxin-3 and Machado-Joseph disease.Prog Neurobiol. 2011 Sep 15;95(1):26-48. doi: 10.1016/j.pneurobio.2011.06.007. Epub 2011 Jun 28. Prog Neurobiol. 2011. PMID: 21740957 Review.
-
Fibrillogenesis of huntingtin and other glutamine containing proteins.Subcell Biochem. 2012;65:225-51. doi: 10.1007/978-94-007-5416-4_10. Subcell Biochem. 2012. PMID: 23225006 Free PMC article. Review.
Cited by
-
TGF-β induces TIAF1 self-aggregation via type II receptor-independent signaling that leads to generation of amyloid β plaques in Alzheimer's disease.Cell Death Dis. 2010 Dec 23;1(12):e110. doi: 10.1038/cddis.2010.83. Cell Death Dis. 2010. PMID: 21368882 Free PMC article.
-
Heparin induces harmless fibril formation in amyloidogenic W7FW14F apomyoglobin and amyloid aggregation in wild-type protein in vitro.PLoS One. 2011;6(7):e22076. doi: 10.1371/journal.pone.0022076. Epub 2011 Jul 13. PLoS One. 2011. PMID: 21779376 Free PMC article.
-
Improvement of neuropathology and transcriptional deficits in CAG 140 knock-in mice supports a beneficial effect of dietary curcumin in Huntington's disease.Mol Neurodegener. 2012 Apr 4;7:12. doi: 10.1186/1750-1326-7-12. Mol Neurodegener. 2012. PMID: 22475209 Free PMC article.
-
Aggregation formation in the polyglutamine diseases: protection at a cost?Mol Cells. 2013 Sep;36(3):185-94. doi: 10.1007/s10059-013-0167-x. Epub 2013 Jun 19. Mol Cells. 2013. PMID: 23794019 Free PMC article. Review.
-
Exploring immunotherapeutic strategies for neurodegenerative diseases: a focus on Huntington's disease and Prion diseases.Acta Pharmacol Sin. 2025 Jun;46(6):1511-1538. doi: 10.1038/s41401-024-01455-w. Epub 2025 Jan 31. Acta Pharmacol Sin. 2025. PMID: 39890942 Review.
References
-
- Uversky VN. Nanoimaging in protein-misfolding and -conformational diseases. Nanomed. 2007;2:615–643. - PubMed
-
- Bates GP. Huntingtin aggregation and toxicity in Huntington's disease. Lancet. 2003;361:1642–1644. - PubMed
-
- Weiss A, Klein C, Woodman B, Sathasivam K, Bibel M, et al. Sensitive biochemical aggregate detection reveals aggregation onset before symptom development in cellular and murine models of Huntington's disease. J Neurochem. 2008;104:846–858. - PubMed
-
- Mangiarini L, Sathasivam K, Seller M, Cozens B, Harper A, et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell. 1996;87:493–506. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

