Differential activation of peritoneal cells by subcutaneous treatment of rats with cryptococcal antigens
- PMID: 19494083
- PMCID: PMC2725545
- DOI: 10.1128/CVI.00100-09
Differential activation of peritoneal cells by subcutaneous treatment of rats with cryptococcal antigens
Abstract
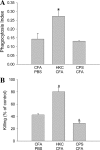
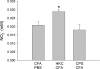
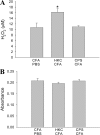
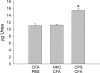
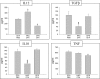
Previous studies in our laboratory have shown that the subcutaneous pretreatment of rats with heat-killed cells (HKC) of Cryptococcus neoformans emulsified in complete Freund adjuvant (CFA) promotes protective immunity against an intraperitoneal challenge with C. neoformans. In contrast, subcutaneous treatment with the capsular polysaccharide (PSC) emulsified in CFA exacerbates the cryptococcal infection. The purpose of this study was to analyze the mechanisms involved in these phenomena. Adherent peritoneal cells from rats treated with HKC-CFA showed upregulated ED2, CD80, and CD86 expression; an increase in the level of production of anticryptococcal metabolites; and the enhanced production of interleukin-12 (IL-12) in comparison with the findings for cells from rats treated with CFA-phosphate-buffered saline (PBS). Adherent peritoneal cells from rats treated with PSC-CFA, however, also presented upregulated ED2, CD80, and CD86 expression compared to the level of expression for peritoneal cells from controls, but these cells showed an increase in arginase activity and decreased levels of production of IL-12 and tumor necrosis factor (TNF) compared with the activity and levels of production by peritoneal cells from CFA-PBS-treated rats. In addition, treatment with HKC-CFA resulted in a rise in the phagocytic and anticryptococcal activities of adherent peritoneal cells compared to those for control rats. However, adherent peritoneal cells from rats treated with PSC-CFA presented a reduction in anticryptococcal activity in comparison with that for cells from animals treated with CFA-PBS. These results show the differential activation between adherent peritoneal cells from HKC-CFA- and PSC-CFA-treated rats, with this differential activation at the primary site of infection possibly being responsible, at least in part, for the phenomena of protection and exacerbation observed in our model.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

