Cellular targeting for cochlear gene therapy
- PMID: 19494575
- PMCID: PMC4379510
- DOI: 10.1159/000218210
Cellular targeting for cochlear gene therapy
Abstract
Gene therapy has considerable potential for the treatment of disorders of the inner ear. Many forms of inherited hearing loss have now been linked to specific locations in the genome, and for many of these the genes and specific mutations involved have been identified. This information provides the basis for therapy based on genetic approaches. However, a major obstacle to gene therapy is the targeting of therapy to the cells and the times that are required. The inner ear is a very complex organ, involving dozens of cell types that must function in a coordinated manner to result in the formation of the ear, and in hearing. Mutations that result in hearing loss can affect virtually any of these cells. Moreover, the genes involved are active during particular times, some for only brief periods of time. In order to be effective, gene therapy must be delivered to the appropriate cells, and at the appropriate times. In many cases, it must also be restricted to these cells and times. This requires methods with which to target gene therapy in space and time. Cell-specific gene promoters offer the opportunity to direct gene therapy to a desired cell type. Moreover, conditional promoters allow gene expression to be turned off and on at desired times. Theoretically, these technologies offer a mechanism by which to deliver gene therapy to any cell, at any given time. This chapter will examine the potential for such targeting to deliver gene therapy to the inner ear in a precisely controlled manner.
Copyright (c) 2009 S. Karger AG, Basel.
Figures

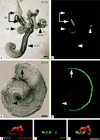

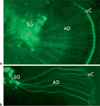
References
-
- Bazan-Peregrino M, Seymour LW, Harris AL. Gene therapy targeting to tumor endothelium. Cancer Gene Ther. 2007;14:117–127. - PubMed
-
- Friedmann T. Gene Therapy: Fact and Fiction in Biology’s New Approaches to Disease. Woodbury: CSHL Press; 1994.
-
- Lodish H, Berk A, Zipursky S, Lawrence S, Matsudaira P, Baltimore D, Darnell J, editors. Molecular Cell Biology. New York: Freeman; 1999.
-
- Verdone L, Agricola E, Caserta M, Di Mauro E. Histone acetylation in gene regulation. Brief Funct Genomic Proteomic. 2006;5:209–221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

