3D architecture of DNA Pol alpha reveals the functional core of multi-subunit replicative polymerases
- PMID: 19494830
- PMCID: PMC2693882
- DOI: 10.1038/emboj.2009.150
3D architecture of DNA Pol alpha reveals the functional core of multi-subunit replicative polymerases
Abstract
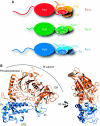
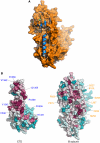
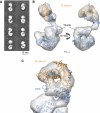
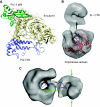
Eukaryotic DNA replication requires the coordinated activity of the multi-subunit DNA polymerases: Pol alpha, Pol delta and Pol epsilon. The conserved catalytic and regulatory B subunits associate in a constitutive heterodimer that represents the functional core of all three replicative polymerases. Here, we combine X-ray crystallography and electron microscopy (EM) to describe subunit interaction and 3D architecture of heterodimeric yeast Pol alpha. The crystal structure of the C-terminal domain (CTD) of the catalytic subunit bound to the B subunit illustrates a conserved mechanism of accessory factor recruitment by replicative polymerases. The EM reconstructions of Pol alpha reveal a bilobal shape with separate catalytic and regulatory modules. Docking of the B-CTD complex in the EM reconstruction shows that the B subunit is tethered to the polymerase domain through a structured but flexible linker. Our combined findings provide a structural template for the common functional architecture of the three major replicative DNA polymerases.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


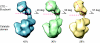
References
-
- Asturias FJ, Cheung IK, Sabouri N, Chilkova O, Wepplo D, Johansson E (2006) Structure of Saccharomyces cerevisiae DNA polymerase epsilon by cryo-electron microscopy. Nat Struct Mol Biol 13: 35–43 - PubMed
-
- Berger I, Fitzgerald DJ, Richmond TJ (2004) Baculovirus expression system for heterologous multiprotein complexes. Nat Biotechnol 22: 1583–1587 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

