Comparison of enzymatic and non-enzymatic means of dissociating adherent monolayers of mesenchymal stem cells
- PMID: 19495917
- PMCID: PMC3055293
- DOI: 10.1007/s12575-009-9001-4
Comparison of enzymatic and non-enzymatic means of dissociating adherent monolayers of mesenchymal stem cells
Abstract
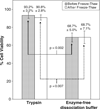
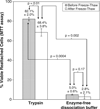
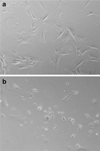
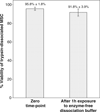
The dissociation of adherent mesenchymal stem cell (MSC) monolayers with trypsin and enzyme-free dissociation buffer was compared. A significantly lower proportion of viable cells were obtained with enzyme-free dissociation buffers compared to trypsin. Subsequently, the dissociated cells were re-seeded on new cell culture dishes and were subjected to the MTT assay 24 h later. The proportion of viable cells that reattached was significantly lower for cells obtained by dissociation with enzyme-free dissociation buffer compared to trypsin. Frozen-thawed MSC displayed a similar trend, yielding consistently higher cell viability and reattachment rates when dissociated with trypsin compared to enzyme-free dissociation buffer. It was also demonstrated that exposure of trypsin-dissociated MSC to enzyme-free dissociation buffer for 1 h had no significant detrimental effect on cell viability.
Figures
Similar articles
-
Effect of Rho-associated kinase (ROCK) inhibitor Y-27632 on the post-thaw viability of cryopreserved human bone marrow-derived mesenchymal stem cells.Tissue Cell. 2009 Oct;41(5):376-80. doi: 10.1016/j.tice.2009.01.004. Epub 2009 Mar 3. Tissue Cell. 2009. PMID: 19261317
-
Effects of Different Cell-Detaching Methods on the Viability and Cell Surface Antigen Expression of Synovial Mesenchymal Stem Cells.Cell Transplant. 2017 Jun 9;26(6):1089-1102. doi: 10.3727/096368917X694831. Epub 2017 Jan 31. Cell Transplant. 2017. PMID: 28139195 Free PMC article.
-
Non-enzymatic dissociation of human mesenchymal stromal cells improves chemokine-dependent migration and maintains immunosuppressive function.Cytotherapy. 2014 Apr;16(4):545-59. doi: 10.1016/j.jcyt.2013.10.003. Cytotherapy. 2014. PMID: 24629709
-
CXCR4 expression by mesenchymal stromal cells is lost after use of enzymatic dissociation agents, but preserved by use of non-enzymatic methods.Int J Hematol. 2021 Jan;113(1):5-9. doi: 10.1007/s12185-020-03043-0. Epub 2021 Jan 3. Int J Hematol. 2021. PMID: 33389659
-
Mechanical dissociation of human embryonic stem cell colonies by manual scraping after collagenase treatment is much more detrimental to cellular viability than is trypsinization with gentle pipetting.Biotechnol Appl Biochem. 2007 May;47(Pt 1):33-7. doi: 10.1042/BA20060151. Biotechnol Appl Biochem. 2007. PMID: 17115976
Cited by
-
Cell Counting and Viability Assessment of 2D and 3D Cell Cultures: Expected Reliability of the Trypan Blue Assay.Biol Proced Online. 2017 Jul 20;19:8. doi: 10.1186/s12575-017-0056-3. eCollection 2017. Biol Proced Online. 2017. PMID: 28814944 Free PMC article.
-
Bioreactor Parameters for Microcarrier-Based Human MSC Expansion under Xeno-Free Conditions in a Vertical-Wheel System.Bioengineering (Basel). 2020 Jul 8;7(3):73. doi: 10.3390/bioengineering7030073. Bioengineering (Basel). 2020. PMID: 32650422 Free PMC article.
-
Proteomic analysis of the extracellular matrix produced by mesenchymal stromal cells: implications for cell therapy mechanism.PLoS One. 2013 Nov 14;8(11):e79283. doi: 10.1371/journal.pone.0079283. eCollection 2013. PLoS One. 2013. PMID: 24244468 Free PMC article.
-
Towards Physiologic Culture Approaches to Improve Standard Cultivation of Mesenchymal Stem Cells.Cells. 2021 Apr 13;10(4):886. doi: 10.3390/cells10040886. Cells. 2021. PMID: 33924517 Free PMC article. Review.
-
Serial Cultivation of an MSC-Like Cell Line with Enzyme-Free Passaging Using a Microporous Titanium Scaffold.Materials (Basel). 2023 Jan 30;16(3):1165. doi: 10.3390/ma16031165. Materials (Basel). 2023. PMID: 36770173 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

