Integrin beta1-focal adhesion kinase signaling directs the proliferation of metastatic cancer cells disseminated in the lungs
- PMID: 19502425
- PMCID: PMC2700942
- DOI: 10.1073/pnas.0904227106
Integrin beta1-focal adhesion kinase signaling directs the proliferation of metastatic cancer cells disseminated in the lungs
Erratum in
- Proc Natl Acad Sci U S A. 2009 Aug 25;106(34):14734
- Proc Natl Acad Sci U S A. 2014 Jan 7;111(1):563
Abstract
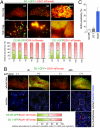
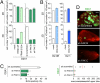
The development of metastases is an extended and inefficient process involving multiple steps. The last of these involves the growth of micrometastases into macroscopic tumors. We show here that intravenously injected, nonmetastatic cancer cells cease proliferating after extravasating into the parenchyma of the lungs; this response is attributable to the cells inability to trigger adhesion-related signaling events when they are scattered sparsely within the extracellular matrix (ECM) of the parenchyma. We recapitulate this situation by culturing these nonmetastatic cells at low seeding density in ECM-derived gels in vitro, in which they undergo cell-cycle arrest resulting, in part, from insufficient activation of focal adhesion kinase (FAK). Metastatic cancer cells, in contrast, show sufficient FAK activation to enable their proliferation within ECM gels in vitro and continue cell-cycle progression within the lung parenchyma in vivo. Activation of FAK in these metastatic cells depends on expression of the beta(1) subunit of integrins, and proliferation of these cells after extravasation in the lungs is diminished by knocking down the expression of either FAK or integrin beta(1). These results demonstrate the critical role of integrin beta(1)-FAK signaling axis in controlling the initial proliferation of micrometastatic cancer cells disseminated in the lungs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Fidler IJ. The pathogenesis of cancer metastasis: The ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. - PubMed
-
- Braun S, et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med. 2005;353:793–802. - PubMed
-
- Mareel MM, De Baetselier P, Roy MV. Mechanism of Invasion and Metastasis. 1st Ed. Boca Raton: CRC press; 1991. p. 584.
-
- Mehlen P, Puisieux A. Metastasis: A question of life or death. Nat Rev Cancer. 2006;6:449–458. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

