Distinct roles for isoforms of the catalytic subunit of class-IA PI3K in the regulation of behaviour of murine embryonic stem cells
- PMID: 19509054
- PMCID: PMC2723149
- DOI: 10.1242/jcs.046557
Distinct roles for isoforms of the catalytic subunit of class-IA PI3K in the regulation of behaviour of murine embryonic stem cells
Abstract
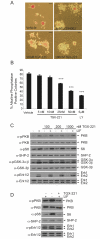
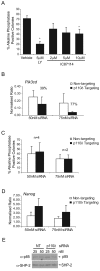
Self-renewal of embryonic stem cells (ESCs) is essential for maintenance of pluripotency, which is defined as the ability to differentiate into any specialised cell type comprising the adult organism. Understanding the mechanisms that regulate ESC self-renewal and proliferation is required before ESCs can fulfil their potential in regenerative therapies, and murine ESCs (mESCs) have been widely used as a model. Members of the class-IA phosphoinositide 3-kinase (PI3K) family of lipid kinases regulate a variety of physiological responses, including cell migration, proliferation and survival. PI3Ks have been reported to regulate both proliferation and self-renewal of mESCs. Here we investigate the contribution of specific class-IA PI3K isoforms to the regulation of mESC fate using small-molecule inhibitors with selectivity for particular class-IA PI3K catalytic isoforms, and siRNA-mediated knockdown. Pharmacological inhibition or knockdown of p110beta promoted mESC differentiation, accompanied by a decrease in expression of Nanog. By comparison, pharmacological inhibition or siRNA-mediated knockdown of p110alpha had no effect on mESC self-renewal per se, but instead appeared to reduce proliferation, which was accompanied by inhibition of leukaemia inhibitory factor (LIF) and insulin-induced PI3K signalling. Our results suggest that PI3Ks contribute to the regulation of both mESC pluripotency and proliferation by differential coupling to selected p110 catalytic isoforms.
Figures





Similar articles
-
Regulation of Nanog expression by phosphoinositide 3-kinase-dependent signaling in murine embryonic stem cells.J Biol Chem. 2007 Mar 2;282(9):6265-73. doi: 10.1074/jbc.M610906200. Epub 2007 Jan 4. J Biol Chem. 2007. PMID: 17204467
-
Regulation of embryonic stem cell self-renewal by phosphoinositide 3-kinase-dependent signaling.J Biol Chem. 2004 Nov 12;279(46):48063-70. doi: 10.1074/jbc.M406467200. Epub 2004 Aug 24. J Biol Chem. 2004. PMID: 15328362
-
Evidence for functional redundancy of class IA PI3K isoforms in insulin signalling.Biochem J. 2007 Jun 15;404(3):449-58. doi: 10.1042/BJ20070003. Biochem J. 2007. PMID: 17362206 Free PMC article.
-
Molecular Mechanisms of Human Disease Mediated by Oncogenic and Primary Immunodeficiency Mutations in Class IA Phosphoinositide 3-Kinases.Front Immunol. 2018 Mar 19;9:575. doi: 10.3389/fimmu.2018.00575. eCollection 2018. Front Immunol. 2018. PMID: 29616047 Free PMC article. Review.
-
Signalling by PI3K isoforms: insights from gene-targeted mice.Trends Biochem Sci. 2005 Apr;30(4):194-204. doi: 10.1016/j.tibs.2005.02.008. Trends Biochem Sci. 2005. PMID: 15817396 Review.
Cited by
-
PI3King the right partner: unique interactions and signaling by p110β.Postdoc J. 2015 Jun;3(6):71-87. doi: 10.14304/surya.jpr.v3n6.8. Postdoc J. 2015. PMID: 26140278 Free PMC article.
-
Disulfiram and BKM120 in Combination with Chemotherapy Impede Tumor Progression and Delay Tumor Recurrence in Tumor Initiating Cell-Rich TNBC.Sci Rep. 2019 Jan 18;9(1):236. doi: 10.1038/s41598-018-35619-6. Sci Rep. 2019. PMID: 30659204 Free PMC article.
-
Runx proteins mediate protective immunity against Leishmania donovani infection by promoting CD40 expression on dendritic cells.PLoS Pathog. 2020 Dec 28;16(12):e1009136. doi: 10.1371/journal.ppat.1009136. eCollection 2020 Dec. PLoS Pathog. 2020. PMID: 33370418 Free PMC article.
-
Pancreatic cancer intrinsic PI3Kα activity accelerates metastasis and rewires macrophage component.EMBO Mol Med. 2021 Jul 7;13(7):e13502. doi: 10.15252/emmm.202013502. Epub 2021 May 25. EMBO Mol Med. 2021. PMID: 34033220 Free PMC article.
-
The crosstalk between long non-coding RNAs and PI3K in cancer.Med Oncol. 2017 Mar;34(3):39. doi: 10.1007/s12032-017-0897-2. Epub 2017 Feb 7. Med Oncol. 2017. PMID: 28176240 Review.
References
-
- Aksoy, I., Sakabedoyan, C., Bourillot, P. Y., Malashicheva, A. B., Mancip, J., Knoblauch, K., Afanassieff, M. and Savatier, P. (2007). Self-renewal of murine embryonic stem cells is supported by the serine/threonine kinases pim-1 and pim-3. Stem Cells 25, 2996-3004. - PubMed
-
- Armstrong, L., Hughes, O., Yung, S., Hyslop, L., Stewart, R., Wappler, I., Peters, H., Walter, T., Stojkovic, P., Evans, J. et al. (2006). The role of PI3K/AKT, MAPK/ERK and NFkappabeta signalling in the maintenance of human embryonic stem cell pluripotency and viability highlighted by transcriptional profiling and functional analysis. Hum. Mol. Gen. 15, 1894-1913. - PubMed
-
- Bi, L., Okabe, I., Bernard, D. J., Wynshaw-Boris, A. and Nussbaum, R. L. (1999). Proliferative defect and embryonic lethality in mice homozygous for a deletion in the p110alpha subunit of phosphoinositide 3-kinase. J. Biol. Chem. 274, 10963-10968. - PubMed
-
- Bi, L., Okabe, I., Bernard, D. J. and Nussbaum, R. L. (2002). Early embryonic lethality in mice deficient in the p110beta catalytic subunit of PI 3-kinase. Mamm. Genome 13, 169-172. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

