Evaluation of Wuchereria bancrofti GST as a vaccine candidate for lymphatic filariasis
- PMID: 19513102
- PMCID: PMC2685978
- DOI: 10.1371/journal.pntd.0000457
Evaluation of Wuchereria bancrofti GST as a vaccine candidate for lymphatic filariasis
Abstract
Background: Lymphatic filarial parasites survive within the lymphatic vessels for years despite the complex immune environment surrounding them. Parasites possibly accomplish this by adopting various immunomodulatory strategies, which include release of glutathione-S-transferases (GSTs) that counteract the oxidative free radicals produced by the host. Since GSTs produced by parasites appear to be critical for the survival of parasites in the host, several studies evaluated the potential of parasite GSTs as vaccine candidates especially against schistosomiasis, fascioliasis and Seteria cervi. However, vaccine potential of GSTs of lymphatic filarial parasites has not been evaluated before.
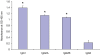
Methods/principal findings: In the present study, the GST gene was cloned from the third stage larval (L3) cDNA libraries of Wuchereria bancrofti, and recombinant GST (WbGST) was expressed and purified. Serum samples from individuals living in an endemic area were analyzed for their reactivity with rWbGST. These findings showed that sera from endemic normal individuals (EN) carry significant levels of anti-WbGST IgG antibodies compared to subjects who are microfilaraemic (Mf) or show symptoms of clinical pathology (CP). Isotype analysis of the anti-WbGST IgG antibodies showed a predominance of IgG1 and IgG3 antibodies in EN individuals. Subsequent functional analysis of the rWbGST showed that the rWbGST protein retained the enzymatic activity of GST and the antibodies in EN sera could inhibit this enzymatic activity. Similar results were obtained when anti-rWbGST antibodies raised in mice were used in the neutralization assay. Brugia malayi GST and WbGST show significant sequence similarity. Therefore, to evaluate the vaccine potential of rWbGST, we used B. malayi L3 as challenge parasites. Vaccine potential of rWbGST was initially evaluated by confirming the role of human and mice WbGST antibodies in an antibody dependent cellular cytotoxicity (ADCC) assay. Subsequent vaccination studies in a jird model showed that approximately 61% protection could be achieved against a B. malayi L3 challenge infection in jirds immunized with rWbGST.
Conclusions: Results of this study show that rWbGST is a potential vaccine candidate against lymphatic filariasis. Nearly 61% protection can be achieved against a B. malayi challenge infection in a jird model. The study also showed that the WbGST protein retained the enzymatic activity of GST and this enzymatic activity appears to be critical for the survival of the parasite in the host.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



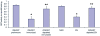

Similar articles
-
Immune response studies with Wuchereria bancrofti vespid allergen homologue (WbVAH) in human lymphatic filariasis.Parasitol Res. 2007 Sep;101(4):981-8. doi: 10.1007/s00436-007-0571-2. Epub 2007 Jun 9. Parasitol Res. 2007. PMID: 17558521 Free PMC article.
-
Humans from Wuchereria bancrofti endemic area elicit substantial immune response to proteins of the filarial parasite Brugia malayi and its endosymbiont Wolbachia.Parasit Vectors. 2017 Jan 24;10(1):40. doi: 10.1186/s13071-016-1963-x. Parasit Vectors. 2017. PMID: 28118850 Free PMC article.
-
Large extracellular loop of tetraspanin as a potential vaccine candidate for filariasis.PLoS One. 2013 Oct 11;8(10):e77394. doi: 10.1371/journal.pone.0077394. eCollection 2013. PLoS One. 2013. PMID: 24146990 Free PMC article.
-
Molecular characterization of antigens of lymphatic filarial parasites.Parasitology. 1986;92 Suppl:S15-38. doi: 10.1017/s003118200008567x. Parasitology. 1986. PMID: 2423945 Review. No abstract available.
-
Advances in immunology and immunopathology of lymphatic filariasis.Southeast Asian J Trop Med Public Health. 1993;24 Suppl 2:76-81. Southeast Asian J Trop Med Public Health. 1993. PMID: 7973952 Review.
Cited by
-
Multivalent fusion protein vaccine for lymphatic filariasis.Vaccine. 2013 Mar 15;31(12):1616-22. doi: 10.1016/j.vaccine.2012.09.055. Epub 2012 Oct 2. Vaccine. 2013. PMID: 23036503 Free PMC article.
-
Advances in Vaccine Development for Human Lymphatic Filariasis.Trends Parasitol. 2020 Feb;36(2):195-205. doi: 10.1016/j.pt.2019.11.005. Epub 2019 Dec 19. Trends Parasitol. 2020. PMID: 31864894 Free PMC article. Review.
-
Harnessing Immune Evasion Strategy of Lymphatic Filariae: A Therapeutic Approach against Inflammatory and Infective Pathology.Vaccines (Basel). 2022 Aug 1;10(8):1235. doi: 10.3390/vaccines10081235. Vaccines (Basel). 2022. PMID: 36016123 Free PMC article. Review.
-
Examining the role of macrolides and host immunity in combatting filarial parasites.Parasit Vectors. 2017 Apr 14;10(1):182. doi: 10.1186/s13071-017-2116-6. Parasit Vectors. 2017. PMID: 28410595 Free PMC article. Review.
-
Troponin 1 of human filarial parasite Brugia malayi: cDNA cloning, expression, purification, and its immunoprophylactic potential.Parasitol Res. 2019 Jun;118(6):1849-1863. doi: 10.1007/s00436-019-06316-8. Epub 2019 May 4. Parasitol Res. 2019. PMID: 31055672
References
-
- Lymphatic filariasis: the disease and its control. Fifth report of the WHO Expert Committee on Filariasis. World Health Organ Tech Rep Ser. 1992;821:1–71. - PubMed
-
- Michael E, Bundy DA, Grenfell BT. Re-assessing the global prevalence and distribution of lymphatic filariasis. Parasitology. 1996;112 (Pt 4):409–428. - PubMed
-
- Fan PC. Diethylcarbamazine treatment of bancroftian and malayan filariasis with emphasis on side effects. Ann Trop Med Parasitol. 1992;86:399–405. - PubMed
-
- Chandrashekar R, Rao UR, Subrahmanyam D. Serum dependent cell-mediated immune reactions to Brugia pahangi infective larvae. Parasite Immunol. 1985;7:633–641. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

