Radical radiotherapy with concurrent weekly cisplatin in loco-regionally advanced squamous cell carcinoma of the head and neck: a single-institution experience
- PMID: 19527507
- PMCID: PMC2702367
- DOI: 10.1186/1758-3284-1-17
Radical radiotherapy with concurrent weekly cisplatin in loco-regionally advanced squamous cell carcinoma of the head and neck: a single-institution experience
Abstract
Background: The dominant pattern of failure for squamous cell carcinoma of head and neck remains loco-regional, although distant metastases are now being increasingly documented. Radical radiotherapy with concurrent chemotherapy is contemporary standard of care in the non-surgical management of these loco-regionally advanced cancers, based on large randomized controlled trials utilizing high-dose cisplatin (80-100 mg/m2) cycled every three-weekly during definitive radiotherapy. Although efficacious, this is associated with high acute morbidity necessitating intensive supportive care with attendant resource implications. The aim of this retrospective study was to assess the efficacy and acute toxicity of an alternative schedule i.e. concurrent weekly cisplatin-based radical radiotherapy and it's potential to be an optimal regimen in advanced head and neck cancers.
Methods: Outcome data of patients with Stage III & IV head and neck squamous cell carcinoma, excluding nasopharynx, planned for radical radiotherapy (66-70 Gy) with concurrent weekly cisplatin (30 mg/m2) treated in a single unit between 1996-2004 was extracted.
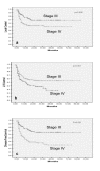
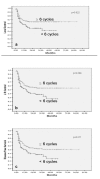
Results: The dataset consisted of 264 patients with a median age of 54 years. The median radiotherapy dose was 70 Gy (range 7.2-72 Gy) and median number of chemotherapy cycles was 6 (range 1-7). Two-thirds (65%) of patients received > or = 85% of planned cisplatin dose. With a mean follow-up of 19 months, the 5-year local control; loco-regional control; and disease free survival was 57%; 46%; and 43% respectively. Acute grade 3 or worse mucositis and dermatitis was seen in 77 (29%) and 92 (35%) patients respectively, essentially in patients receiving doses > or = 66 Gy and 6 or more cycles of chemotherapy. Other toxicities (hematologic, nausea and vomiting) were mild and self-limiting. Overall, the acute toxicity of this concurrent weekly chemo-radiation regimen though mildly increased did not mandate intensive supportive care. Stage grouping, primary site, and intensity of treatment were significant predictors of loco-regional control and disease free survival.
Conclusion: Radical radiotherapy with concurrent weekly cisplatin has moderate efficacy and acceptable acute toxicity with potential to be an optimal regimen in loco-regionally advanced squamous cell carcinoma of the head and neck, particularly in limited-resource settings. Stage grouping, primary site, and treatment intensity are important determinants of outcome.
Figures

Similar articles
-
Conventional radiotherapy with concurrent weekly Cisplatin in locally advanced head and neck cancers of squamous cell origin - a single institution experience.Asian Pac J Cancer Prev. 2013;14(11):6883-8. doi: 10.7314/apjcp.2013.14.11.6883. Asian Pac J Cancer Prev. 2013. PMID: 24377620 Clinical Trial.
-
Concurrent chemo-irradiation using accelerated concomitant boost radiation therapy in loco-regionally advanced head and neck squamous cell carcinomas.J Cancer Res Ther. 2006 Jul-Sep;2(3):90-6. doi: 10.4103/0973-1482.27582. J Cancer Res Ther. 2006. PMID: 17998685 Clinical Trial.
-
Phase II/III Randomized Controlled Trial of Concomitant Hyperfractionated Radiotherapy plus Cetuximab (Anti-EGFR Antibody) or Chemotherapy in Locally Advanced Head and Neck Cancer.Gulf J Oncolog. 2019 May;1(30):6-12. Gulf J Oncolog. 2019. PMID: 31242976 Clinical Trial.
-
Hyperfractionated radiotherapy with concurrent chemotherapy for para-aortic lymph node recurrence in carcinoma of the cervix.Int J Radiat Oncol Biol Phys. 2003 Apr 1;55(5):1247-53. doi: 10.1016/s0360-3016(02)04401-2. Int J Radiat Oncol Biol Phys. 2003. PMID: 12654434 Review.
-
Resource-sparing curative-intent hypofractionated-accelerated radiotherapy in head and neck cancer: More relevant than ever before in the COVID era.Oral Oncol. 2020 Dec;111:105045. doi: 10.1016/j.oraloncology.2020.105045. Epub 2020 Oct 20. Oral Oncol. 2020. PMID: 33091846 Free PMC article.
Cited by
-
Squamous cell carcinoma metastatic to cervical lymph nodes from unknown primary origin: the impact of chemoradiotherapy.Chin J Cancer. 2012 Oct;31(10):484-90. doi: 10.5732/cjc.012.10035. Epub 2012 Jun 6. Chin J Cancer. 2012. PMID: 22692070 Free PMC article.
-
The comparison between weekly and three-weekly cisplatin delivered concurrently with radiotherapy for patients with postoperative high-risk squamous cell carcinoma of the oral cavity.Radiat Oncol. 2012 Dec 18;7:215. doi: 10.1186/1748-717X-7-215. Radiat Oncol. 2012. PMID: 23245290 Free PMC article. Clinical Trial.
-
Trolamine emulsion for the prevention of radiation dermatitis in patients with squamous cell carcinoma of the head and neck.Support Care Cancer. 2012 Jan;20(1):185-90. doi: 10.1007/s00520-011-1110-3. Epub 2011 Feb 22. Support Care Cancer. 2012. PMID: 21340657 Clinical Trial.
-
Organ preservation strategies: Review of literature and their applicability in developing nations.South Asian J Cancer. 2014 Jul;3(3):147-50. doi: 10.4103/2278-330X.136764. South Asian J Cancer. 2014. PMID: 25136519 Free PMC article. Review.
-
A prospective randomized study comparing concurrent chemoradiation with weekly and 3 weekly cisplatin in locally advanced oropharyngeal carcinoma.South Asian J Cancer. 2019 Jul-Sep;8(3):178-182. doi: 10.4103/sajc.sajc_270_18. South Asian J Cancer. 2019. PMID: 31489293 Free PMC article.
References
-
- GLOBOCAN 2002 . Version 20 IARC Cancer Base No5. Lyon: IARC Press; 2004. Cancer incidence, mortality, and prevalence worldwide.
-
- Pignon JP, Bourhis J, Domenge C, Designe L. Chemotherapy added to locoregional treatment for head and neck squamous-cell carcinoma: three meta-analyses of updated individual data. Lancet. 2000;355:949–955. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

