Dysfunction of the dentate basket cell circuit in a rat model of temporal lobe epilepsy
- PMID: 19535596
- PMCID: PMC2838908
- DOI: 10.1523/JNEUROSCI.6199-08.2009
Dysfunction of the dentate basket cell circuit in a rat model of temporal lobe epilepsy
Abstract
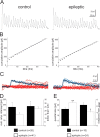
Temporal lobe epilepsy is common and difficult to treat. Reduced inhibition of dentate granule cells may contribute. Basket cells are important inhibitors of granule cells. Excitatory synaptic input to basket cells and unitary IPSCs (uIPSCs) from basket cells to granule cells were evaluated in hippocampal slices from a rat model of temporal lobe epilepsy. Basket cells were identified by electrophysiological and morphological criteria. Excitatory synaptic drive to basket cells, measured by mean charge transfer and frequency of miniature EPSCs, was significantly reduced after pilocarpine-induced status epilepticus and remained low in epileptic rats, despite mossy fiber sprouting. Paired recordings revealed higher failure rates and a trend toward lower amplitude uIPSCs at basket cell-to-granule cell synapses in epileptic rats. Higher failure rates were not attributable to excessive presynaptic inhibition of GABA release by activation of muscarinic acetylcholine or GABA(B) receptors. High-frequency trains of action potentials in basket cells generated uIPSCs in granule cells to evaluate readily releasable pool (RRP) size and resupply rate of recycling vesicles. Recycling rate was similar in control and epileptic rats. However, quantal size at basket cell-to-granule cell synapses was larger and RRP size smaller in epileptic rats. Therefore, in epileptic animals, basket cells receive less excitatory synaptic drive, their pools of readily releasable vesicles are smaller, and transmission failure at basket cell-to-granule cell synapses is increased. These findings suggest dysfunction of the dentate basket cell circuit could contribute to hyperexcitability and seizures.
Figures


References
-
- Andrioli A, Alonso-Nanclares L, Arellano JI, DeFelipe J. Quantitative analysis of parvalbumin-immunoreactive cells in the human epileptic hippocampus. Neuroscience. 2007;149:131–143. - PubMed
-
- Austin JE, Buckmaster PS. Recurrent excitation of granule cells with basal dendrites and low interneuron density and inhibitory postsynaptic current frequency in the dentate gyrus of macaque monkeys. J Comp Neurol. 2004;476:205–218. - PubMed
-
- Babb TL, Kupfer WR, Pretorius JK, Crandall PH, Levesque MF. Synaptic reorganization by mossy fibers in human epileptic fascia dentata. Neuroscience. 1991;42:351–353. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
