Aging and human CD4(+) regulatory T cells
- PMID: 19540259
- PMCID: PMC2753872
- DOI: 10.1016/j.mad.2009.06.003
Aging and human CD4(+) regulatory T cells
Abstract
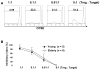
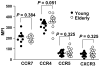
Alterations in immunity that occur with aging likely contribute to the development of infection, malignancy and inflammatory diseases. Naturally occurring CD4(+) regulatory T cells (Treg) expressing high levels of CD25 and forkhead box P3 (FOXP3) are essential for regulating immune responses. Here we investigated the effect of aging on the number, phenotypes and function of CD4(+) Treg in humans. The frequency and phenotypic characteristics of CD4(+), FOXP3(+) T cells as well as their capacity to suppress inflammatory cytokine production and proliferation of CD4(+), CD25(-) T cells (target cells) were comparable in young (age <or=40) and elderly (age >or=65) individuals. However, when CD4(+), FOXP3(+) Treg and CD4(+), CD25(-) T cells were co-cultured at a ratio of 1:1, the production of anti-inflammatory cytokine IL-10 from CD4(+), CD25(-) T cells was more potently suppressed in the elderly than in the young. This finding was not due to changes in CTLA-4 expression or apoptosis of CD4(+), FOXP3(+) Treg and CD4(+), CD25(-) T cells. Taken together, our observations suggest that aging may affect the capacity of CD4(+), FOXP3(+) T cells in regulating IL-10 production from target CD4(+) T cells in humans although their other cellular characteristics remain unchanged.
Figures




Similar articles
-
Plasmodium falciparum-mediated induction of human CD25Foxp3 CD4 T cells is independent of direct TCR stimulation and requires IL-2, IL-10 and TGFbeta.PLoS Pathog. 2009 Aug;5(8):e1000543. doi: 10.1371/journal.ppat.1000543. Epub 2009 Aug 14. PLoS Pathog. 2009. PMID: 19680449 Free PMC article.
-
IL-1R1 is expressed on both Helios(+) and Helios(-) FoxP3(+) CD4(+) T cells in the rheumatic joint.Clin Exp Immunol. 2015 Oct;182(1):90-100. doi: 10.1111/cei.12668. Epub 2015 Jul 30. Clin Exp Immunol. 2015. PMID: 26076982 Free PMC article.
-
Stimulation of α7 nicotinic acetylcholine receptor by nicotine increases suppressive capacity of naturally occurring CD4+CD25+ regulatory T cells in mice in vitro.J Pharmacol Exp Ther. 2010 Dec;335(3):553-61. doi: 10.1124/jpet.110.169961. Epub 2010 Sep 15. J Pharmacol Exp Ther. 2010. PMID: 20843956
-
Critical stoichiometric ratio of CD4(+) CD25(+) FoxP3(+) regulatory T cells and CD4(+) CD25(-) responder T cells influence immunosuppression in patients with B-cell acute lymphoblastic leukaemia.Immunology. 2014 May;142(1):124-39. doi: 10.1111/imm.12237. Immunology. 2014. PMID: 24354800 Free PMC article.
-
Equine CD4(+) CD25(high) T cells exhibit regulatory activity by close contact and cytokine-dependent mechanisms in vitro.Immunology. 2011 Nov;134(3):292-304. doi: 10.1111/j.1365-2567.2011.03489.x. Immunology. 2011. PMID: 21977999 Free PMC article.
Cited by
-
An altered relationship of influenza vaccine-specific IgG responses with T cell immunity occurs with aging in humans.Clin Immunol. 2013 May;147(2):79-88. doi: 10.1016/j.clim.2013.02.022. Epub 2013 Mar 15. Clin Immunol. 2013. PMID: 23578549 Free PMC article.
-
Diminished Pneumococcal-Specific CD4+ T-Cell Response is Associated With Increased Regulatory T Cells at Older Age.Front Aging. 2021 Nov 3;2:746295. doi: 10.3389/fragi.2021.746295. eCollection 2021. Front Aging. 2021. PMID: 35822055 Free PMC article.
-
T Cell Aging in Patients with Colorectal Cancer-What Do We Know So Far?Cancers (Basel). 2021 Dec 11;13(24):6227. doi: 10.3390/cancers13246227. Cancers (Basel). 2021. PMID: 34944847 Free PMC article. Review.
-
Phenotypic complexity of T regulatory subsets in patients with B-chronic lymphocytic leukemia.Mod Pathol. 2012 Feb;25(2):246-59. doi: 10.1038/modpathol.2011.164. Epub 2011 Nov 18. Mod Pathol. 2012. PMID: 22101351 Free PMC article.
-
Regulatory T cells in induced sputum of asthmatic children: association with inflammatory cytokines.Multidiscip Respir Med. 2010 Feb 28;5(1):22-30. doi: 10.1186/2049-6958-5-1-22. Multidiscip Respir Med. 2010. PMID: 22958596 Free PMC article.
References
-
- Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. - PubMed
-
- Baecher-Allan C, Brown JA, Freeman GJ, Hafler DA. CD4+CD25high regulatory cells in human peripheral blood. J Immunol. 2001;167:1245–1253. - PubMed
-
- Bennett CL, Christie J, Ramsdell F, Brunkow ME, Ferguson PJ, Whitesell L, Kelly TE, Saulsbury FT, Chance PF, Ochs HD. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20–21. - PubMed
-
- Brunkow ME, Jeffery EW, Hjerrild KA, Paeper B, Clark LB, Yasayko SA, Wilkinson JE, Galas D, Ziegler SF, Ramsdell F. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet. 2001;27:68–73. - PubMed
-
- Chorinchath BB, Kong LY, Mao L, McCallum RE. Age-associated differences in TNF-alpha and nitric oxide production in endotoxic mice. J Immunol. 1996;156:1525–1530. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

