Structural and functional characterization of the interdomain interaction in the mineralocorticoid receptor
- PMID: 19541744
- PMCID: PMC2737563
- DOI: 10.1210/me.2009-0032
Structural and functional characterization of the interdomain interaction in the mineralocorticoid receptor
Abstract
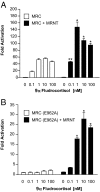
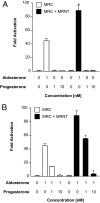
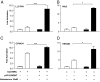
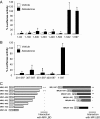
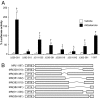
The mineralocorticoid receptor (MR) plays a central role in electrolyte homeostasis and in cardiovascular disease. We have previously reported a ligand-dependent N/C-interaction in the MR. In the present study we sought to fully characterize the MR N/C-interaction. By using a range of natural and synthetic MR ligands in a mammalian two-hybrid assay we demonstrate that in contrast to aldosterone, which strongly induces the interaction, the physiological ligands deoxycorticosterone and cortisol weakly promote the interaction but predominantly inhibit the aldosterone-mediated N/C-interaction. Similarly, progesterone and dexamethasone antagonize the interaction. In contrast, the synthetic agonist 9alpha-fludrocortisol robustly induces the interaction. The ability of the N/C interaction to discriminate between MR agonists suggests a subtle conformational difference in the ligand-binding domain induced by these agonists. We also demonstrate that the N/C interaction is not cell specific, consistent with the evidence from a glutathione-S-transferase pull-down assay, of a direct protein-protein interaction between the N- and C-terminal domains of the MR. Examination of a panel of deletions in the N terminus suggests that several regions may be critical to the N/C-interaction. These studies have identified functional differences between physiological MR ligands, which suggest that the ligand-specific dependence of the N/C-interaction may contribute to the differential activation of the MR that has been reported in vivo.
Figures


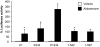

Similar articles
-
Interdomain interactions in the mineralocorticoid receptor.Mol Cell Endocrinol. 2003 Feb 28;200(1-2):45-55. doi: 10.1016/s0303-7207(02)00413-6. Mol Cell Endocrinol. 2003. PMID: 12644298
-
NF-YC functions as a corepressor of agonist-bound mineralocorticoid receptor.J Biol Chem. 2010 Mar 12;285(11):8084-93. doi: 10.1074/jbc.M109.053371. Epub 2010 Jan 6. J Biol Chem. 2010. PMID: 20054001 Free PMC article.
-
Mechanisms of ligand specificity of the mineralocorticoid receptor.J Endocrinol. 2012 Apr;213(1):15-24. doi: 10.1530/JOE-11-0372. Epub 2011 Dec 12. J Endocrinol. 2012. PMID: 22159507 Review.
-
Identification and characterization of a ligand-selective mineralocorticoid receptor coactivator.FASEB J. 2014 Oct;28(10):4200-10. doi: 10.1096/fj.13-242479. Epub 2014 Jun 6. FASEB J. 2014. PMID: 24907116
-
Novel interactions of the mineralocorticoid receptor.Mol Cell Endocrinol. 2015 Jun 15;408:33-7. doi: 10.1016/j.mce.2015.01.027. Epub 2015 Feb 7. Mol Cell Endocrinol. 2015. PMID: 25662276 Review.
Cited by
-
Aldosterone: Renal Action and Physiological Effects.Compr Physiol. 2023 Mar 30;13(2):4409-4491. doi: 10.1002/cphy.c190043. Compr Physiol. 2023. PMID: 36994769 Free PMC article.
-
A tumour suppressive relationship between mineralocorticoid and retinoic acid receptors activates a transcriptional program consistent with a reverse Warburg effect in breast cancer.Breast Cancer Res. 2020 Nov 4;22(1):122. doi: 10.1186/s13058-020-01355-x. Breast Cancer Res. 2020. PMID: 33148314 Free PMC article.
-
Estrogen receptor inhibits mineralocorticoid receptor transcriptional regulatory function.Endocrinology. 2014 Nov;155(11):4461-72. doi: 10.1210/en.2014-1270. Epub 2014 Jul 22. Endocrinology. 2014. PMID: 25051445 Free PMC article.
-
Glucocorticoid receptor Thr524 phosphorylation by MINK1 induces interactions with 14-3-3 protein regulators.J Biol Chem. 2021 Jan-Jun;296:100551. doi: 10.1016/j.jbc.2021.100551. Epub 2021 Mar 17. J Biol Chem. 2021. PMID: 33744286 Free PMC article.
-
Analysis of interdomain interactions of the androgen receptor.Methods Mol Biol. 2011;776:113-29. doi: 10.1007/978-1-61779-243-4_8. Methods Mol Biol. 2011. PMID: 21796524 Free PMC article.
References
-
- Rogerson FM, Fuller PJ 2000 Mineralocorticoid action. Steroids 65:61–73 - PubMed
-
- Farman N, Rafestin-Oblin ME 2001 Multiple aspects of mineralocorticoid selectivity. Am J Physiol Renal Physiol 280:F181–F192 - PubMed
-
- Cachofeiro V, Miana M, de Las Heras N, Martín-Fernández B, Ballesteros S, Fernández-Tresguerres J, Lahera V 2008 Aldosterone and the vascular system. J Steroid Biochem Mol Biol 109:331–335 - PubMed
-
- Struthers AD 2004 Aldosterone in heart failure: pathophysiology and treatment. Curr Heart Fail Rep 1:171–175 - PubMed
-
- Fuller PJ, Young MJ 2005 Mechanisms of mineralocorticoid action. Hypertension 46:1227–1235 - PubMed

