Modulation of TGFbeta1-dependent myofibroblast differentiation by hyaluronan
- PMID: 19541937
- PMCID: PMC2708802
- DOI: 10.2353/ajpath.2009.080837
Modulation of TGFbeta1-dependent myofibroblast differentiation by hyaluronan
Abstract
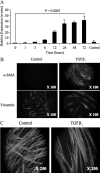
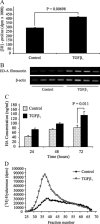
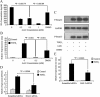
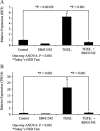
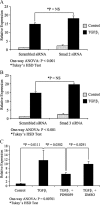
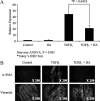
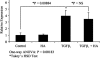
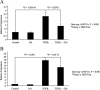
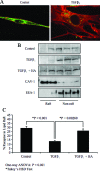
Myofibroblasts are contractile cells that are characterized by the expression of alpha-smooth muscle actin and mediate the closure of wounds and the formation of collagen-rich scars. Their presence in organs such as lungs, liver, and kidney has long been established as a marker of progressive fibrosis. The transforming growth factor beta(1)-driven differentiation of fibroblasts is a major source of myofibroblasts, and recent data have shown that hyaluronan is a major modulator of this process. This study examines this differentiation mechanism in more detail. Transforming growth factor beta(1)-dependent differentiation to the myofibroblastic phenotype was antagonized by the inhibition of hyaluronan synthesis, confirming that hyaluronan was necessary for differentiation. This response, however, was not reproduced by simply adding hyaluronan to fibroblasts, as the results implicated hyaladherins, as well as the macromolecular assembly of de novo hyaluronan, as essential in this process. We previously suggested that there is a relocalization of lipid-raft components during myofibroblastic differentiation. The present study demonstrates that the hyaluronan receptor CD44, the hyaluronidase HYAL 2, and the transforming growth factor beta(1)-receptor ALK5 all relocalized from raft to non-raft locations, which was reversed by the addition of exogenous hyaluronan. These data highlight a role for endogenous hyaluronan in the mediation of myofibroblastic differentiation. While hyaluronan synthesis was both essential and necessary for differentiation, exogenously provided hyaluronan antagonized differentiation, underscoring a pathological role for hyaluronan in such cell fate processes.
Figures

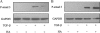
Similar articles
-
Transforming growth factor-β1 (TGF-β1)-stimulated fibroblast to myofibroblast differentiation is mediated by hyaluronan (HA)-facilitated epidermal growth factor receptor (EGFR) and CD44 co-localization in lipid rafts.J Biol Chem. 2013 May 24;288(21):14824-38. doi: 10.1074/jbc.M113.451336. Epub 2013 Apr 15. J Biol Chem. 2013. PMID: 23589287 Free PMC article.
-
Tumor Necrosis Factor-stimulated Gene 6 (TSG-6)-mediated Interactions with the Inter-α-inhibitor Heavy Chain 5 Facilitate Tumor Growth Factor β1 (TGFβ1)-dependent Fibroblast to Myofibroblast Differentiation.J Biol Chem. 2016 Jun 24;291(26):13789-801. doi: 10.1074/jbc.M115.670521. Epub 2016 May 3. J Biol Chem. 2016. PMID: 27143355 Free PMC article.
-
Dysfunctional pericellular hyaluronan deposition contributes to attenuated CD44/EGFR co-localization and impaired myofibroblast differentiation in chronic wound fibroblasts.Exp Cell Res. 2025 Jul 15;450(2):114646. doi: 10.1016/j.yexcr.2025.114646. Epub 2025 Jun 14. Exp Cell Res. 2025. PMID: 40523596
-
Age-related changes in pericellular hyaluronan organization leads to impaired dermal fibroblast to myofibroblast differentiation.Am J Pathol. 2009 Nov;175(5):1915-28. doi: 10.2353/ajpath.2009.090045. Epub 2009 Oct 1. Am J Pathol. 2009. PMID: 19808648 Free PMC article.
-
Myofibroblasts: Function, Formation, and Scope of Molecular Therapies for Skin Fibrosis.Biomolecules. 2021 Jul 23;11(8):1095. doi: 10.3390/biom11081095. Biomolecules. 2021. PMID: 34439762 Free PMC article. Review.
Cited by
-
A RHAMM mimetic peptide blocks hyaluronan signaling and reduces inflammation and fibrogenesis in excisional skin wounds.Am J Pathol. 2012 Oct;181(4):1250-70. doi: 10.1016/j.ajpath.2012.06.036. Epub 2012 Aug 11. Am J Pathol. 2012. PMID: 22889846 Free PMC article.
-
Transforming Growth Factor-β Receptor-Mediated, p38 Mitogen-Activated Protein Kinase-Dependent Signaling Drives Enhanced Myofibroblast Differentiation during Skin Wound Healing in Mice Lacking Hyaluronan Synthases 1 and 3.Am J Pathol. 2022 Dec;192(12):1683-1698. doi: 10.1016/j.ajpath.2022.08.003. Epub 2022 Sep 3. Am J Pathol. 2022. PMID: 36063901 Free PMC article.
-
Aberrant mural cell recruitment to lymphatic vessels and impaired lymphatic drainage in a murine model of pulmonary fibrosis.Blood. 2012 Jun 14;119(24):5931-42. doi: 10.1182/blood-2011-12-396895. Epub 2012 Apr 30. Blood. 2012. PMID: 22547584 Free PMC article.
-
Esophageal Squamous Cell Carcinoma Cells Modulate Chemokine Expression and Hyaluronan Synthesis in Fibroblasts.J Biol Chem. 2016 Feb 19;291(8):4091-106. doi: 10.1074/jbc.M115.708909. Epub 2015 Dec 23. J Biol Chem. 2016. PMID: 26699196 Free PMC article.
-
Antiadhesive Hyaluronic Acid-Based Wound Dressings Promote Wound Healing by Preventing Re-Injury: An In Vivo Investigation.Biomedicines. 2024 Feb 23;12(3):510. doi: 10.3390/biomedicines12030510. Biomedicines. 2024. PMID: 38540123 Free PMC article.
References
-
- Gabbiani G. The myofibroblast in wound healing and fibrocontractive diseases. J Pathol. 2003;200:500–503. - PubMed
-
- Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol. 2002;3:349–363. - PubMed
-
- Desmouliere A, Darby IA, Gabbiani G. Normal and pathologic soft tissue remodeling: role of the myofibroblast, with special emphasis on liver and kidney fibrosis. Lab Invest. 2003;83:1689–1707. - PubMed
-
- Hinz B, Gabbiani G. Cell-matrix and cell-cell contacts of myofibroblasts: role in connective tissue remodeling. Thromb Haemost. 2003;90:993–1002. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

