Peroxiredoxin Tsa1 is the key peroxidase suppressing genome instability and protecting against cell death in Saccharomyces cerevisiae
- PMID: 19543365
- PMCID: PMC2688748
- DOI: 10.1371/journal.pgen.1000524
Peroxiredoxin Tsa1 is the key peroxidase suppressing genome instability and protecting against cell death in Saccharomyces cerevisiae
Abstract
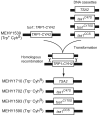
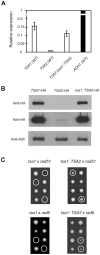
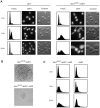
Peroxiredoxins (Prxs) constitute a family of thiol-specific peroxidases that utilize cysteine (Cys) as the primary site of oxidation during the reduction of peroxides. To gain more insight into the physiological role of the five Prxs in budding yeast Saccharomyces cerevisiae, we performed a comparative study and found that Tsa1 was distinguished from the other Prxs in that by itself it played a key role in maintaining genome stability and in sustaining aerobic viability of rad51 mutants that are deficient in recombinational repair. Tsa2 and Dot5 played minor but distinct roles in suppressing the accumulation of mutations in cooperation with Tsa1. Tsa2 was capable of largely complementing the absence of Tsa1 when expressed under the control of the Tsa1 promoter. The presence of peroxidatic cysteine (Cys(47)) was essential for Tsa1 activity, while Tsa1(C170S) lacking the resolving Cys was partially functional. In the absence of Tsa1 activity (tsa1 or tsa1(CCS) lacking the peroxidatic and resolving Cys) and recombinational repair (rad51), dying cells displayed irregular cell size/shape, abnormal cell cycle progression, and significant increase of phosphatidylserine externalization, an early marker of apoptosis-like cell death. The tsa1(CCS) rad51- or tsa1 rad51-induced cell death did not depend on the caspase Yca1 and Ste20 kinase, while the absence of the checkpoint protein Rad9 accelerated the cell death processes. These results indicate that the peroxiredoxin Tsa1, in cooperation with appropriate DNA repair and checkpoint mechanisms, acts to protect S. cerevisiae cells against toxic levels of DNA damage that occur during aerobic growth.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Rhee SG, Chae HZ, Kim K. Peroxiredoxins: a historical overview and speculative preview of novel mechanisms and emerging concepts in cell signaling. Free Radic Biol Med. 2005;38:1543–1552. - PubMed
-
- Wood ZA, Schroder E, Robin Harris J, Poole LB. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem Sci. 2003;28:32–40. - PubMed
-
- Park SG, Cha MK, Jeong W, Kim IH. Distinct physiological functions of thiol peroxidase isoenzymes in Saccharomyces cerevisiae. J Biol Chem. 2000;275:5723–5732. - PubMed
-
- Poole LB. Bacterial defenses against oxidants: mechanistic features of cysteine-based peroxidases and their flavoprotein reductases. Arch Biochem Biophys. 2005;433:240–254. - PubMed
-
- Biteau B, Labarre J, Toledano MB. ATP-dependent reduction of cysteine-sulphinic acid by S. cerevisiae sulphiredoxin. Nature. 2003;425:980–984. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

