Nucleotide analogue prodrug tenofovir disoproxil enhances lymphoid cell loading following oral administration in monkeys
- PMID: 19545170
- PMCID: PMC2777719
- DOI: 10.1021/mp900036s
Nucleotide analogue prodrug tenofovir disoproxil enhances lymphoid cell loading following oral administration in monkeys
Abstract
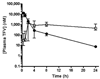
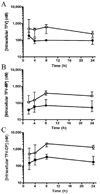
The antiviral drug tenofovir (TFV) is orally administered as the fumarate salt of its disoproxil prodrug (TFV disoproxil fumarate (TDF)). TFV is a dianion at physiological pH and, as a result, has poor lipid membrane permeability. Administration of the lipophilic and cell-permeable prodrug, TFV disoproxil, enhances the oral absorption of TFV. In order to determine whether oral administration of TDF also increases distribution to sites of viral infection, the plasma and circulating lymphoid cell pharmacokinetics of TFV and its phosphorylated metabolites were assessed following a single oral TDF or subcutaneous TFV administration at doses yielding equivalent plasma exposures to TFV in macaques. Despite TFV disoproxil's lack of plasma stability and undetectable levels in the first plasma samples taken, oral administration of TDF resulted in 7.9-fold higher peripheral blood mononuclear cell exposures to the active metabolite, TFV-diphosphate. The apparent plasma terminal half-life (t(1/2)) of TFV was also longer following oral TDF relative to subcutaneous TFV administration (median t(1/2) of 15.3 and 3.9 h, respectively), suggesting broader distribution to cells and tissues outside of the central plasma compartment. In conclusion, the disoproxil pro-moiety enhances not only the oral absorption of TFV but also tissue and lymphoid cell loading. These results illustrate that administration of even a fleeting prodrug can increase target tissue loading and give valuable insight for future prodrug development.
Figures

References
-
- Robbins BL, Srinivas RV, Kim C, Bischofberger N, Fridland A. Anti-human immunodeficiency virus activity and cellular metabolism of a potential prodrug of the acyclic nucleoside phosphonate 9-R-(2-phosphonomethoxypropyl)adenine (PMPA), Bis(isopropyloxymethylcarbonyl)PMPA. Antimicrob. Agents Chemother. 1998;42:612–617. - PMC - PubMed
-
- Barditch-Crovo P, Deeks SG, Collier A, Safrin S, Coakley DF, Miller M, Kearney BP, Coleman RL, Lamy PD, Kahn JO, McGowan I, Lietman PS. Phase i/ii trial of the pharmacokinetics, safety, and antiretroviral activity of tenofovir disoproxil fumarate in human immunodeficiency virus-infected adults. Antimicrob. Agents Chemother. 2001;45:2733–2739. - PMC - PubMed
-
- Kearney BP, Flaherty JF, Shah J. Tenofovir disoproxil fumarate: clinical pharmacology and pharmacokinetics. Clin. Pharmacokinet. 2004;43:595–612. - PubMed
-
- Lee WA, He GX, Eisenberg E, Cihlar T, Swaminathan S, Mulato A, Cundy KC. Selective intracellular activation of a novel prodrug of the human immunodeficiency virus reverse transcriptase inhibitor tenofovir leads to preferential distribution and accumulation in lymphatic tissue. Antimicrob. Agents Chemother. 2005;49:1898–1906. - PMC - PubMed
-
- van Gelder J, Deferme S, Naesens L, De Clercq E, van den Mooter G, Kinget R, Augustijns P. Intestinal absorption enhancement of the ester prodrug tenofovir disoproxil fumarate through modulation of the biochemical barrier by defined ester mixtures. Drug Metab. Dispos. 2002;30:924–930. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

