Electrophysiological properties of thalamic, subthalamic and nigral neurons during the anti-parkinsonian placebo response
- PMID: 19546163
- PMCID: PMC2746615
- DOI: 10.1113/jphysiol.2009.169425
Electrophysiological properties of thalamic, subthalamic and nigral neurons during the anti-parkinsonian placebo response
Abstract
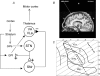
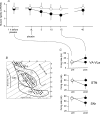
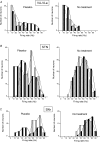
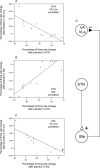
Placebo administration to Parkinson patients is known to induce dopamine release in the striatum and to affect the activity of subthalamic nucleus (STN) neurons. By using intraoperative single-neuron recording techniques in awake patients, here we extend our previous study on STN recording, and characterize part of the neuronal circuit which is affected by placebos. In those patients who showed a clinical placebo response, there was a decrease in firing rate in STN neurons that was associated with a decrease in the substantia nigra pars reticulata (SNr) and an increase in the ventral anterior (VA) and anterior ventral lateral (VLa) thalamus. These data show that placebo decreases STN and SNr activity whereas it increases VA/VLa activity. By contrast, placebo non-responders showed either a lack of changes in this circuit or partial changes in the STN only. Thus, changes in activity in the whole basal ganglia-VA/VLa circuit appear to be important in order to observe a clinical placebo improvement, although the involvement of other circuits, such as the direct pathway bypassing the STN, cannot be ruled out. The circuit we describe in the present study is likely to be a part of a more complex circuitry, including the striatum and the internal globus pallidus (GPi), that is modified by placebo administration. These findings indicate that a placebo treatment, which is basically characterized by verbal suggestions of benefit, can reverse the malfunction of a complex neuronal circuit, although these placebo-associated neuronal changes are short-lasting and occur only in some patients but not in others.
Figures

References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Benarroch EE. Subthalamic nucleus and its connections: Anatomic substrate for the network effects of deep brain stimulation. Neurology. 2008;70:1991–1995. - PubMed
-
- Benazzouz A, Gao DM, Ni ZG, Piallat B, Bouali-Benazzouz R, Benabid AL. Effect of high-frequency stimulation of the subthalamic nucleus on the neuronal activities of the substantia nigra pars reticulata and ventrolateral nucleus of the thalamus in the rat. Neuroscience. 2000;99:289–295. - PubMed
-
- Benazzouz A, Hallett M. Mechanism of action of deep brain stimulation. Neurology. 2000;55(Suppl. 6):S13–S17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

