In situ analysis of protein S-glutathionylation in lung tissue using glutaredoxin-1-catalyzed cysteine derivatization
- PMID: 19556513
- PMCID: PMC2708792
- DOI: 10.2353/ajpath.2009.080736
In situ analysis of protein S-glutathionylation in lung tissue using glutaredoxin-1-catalyzed cysteine derivatization
Abstract
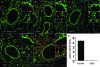
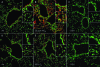
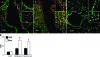
Protein S-glutathionylation (PSSG) is a posttranslational modification that involves the conjugation of the small antioxidant molecule glutathione to cysteine residues and is emerging as a critical mechanism of redox-based signaling. PSSG levels increase under conditions of oxidative stress and are controlled by glutaredoxins (Grx) that, under physiological conditions, preferentially deglutathionylate cysteines and restore sulfhydryls. Both the occurrence and distribution of PSSG in tissues is unknown because of the labile nature of this oxidative event and the lack of specific reagents. The goal of this study was to establish and validate a protocol that enables detection of PSSG in situ, using the property of Grx to deglutathionylate cysteines. Using Grx1-catalyzed cysteine derivatization, we evaluated PSSG content in mice subjected to various models of lung injury and fibrosis. In control mice, PSSG was detectable primarily in the airway epithelium and alveolar macrophages. Exposure of mice to NO(2) resulted in enhanced PSSG levels in parenchymal regions, while exposure to O(2) resulted in minor detectable changes. Finally, bleomycin exposure resulted in marked increases in PSSG reactivity both in the bronchial epithelium as well as in parenchymal regions. Taken together, these findings demonstrate that Grx1-based cysteine derivatization is a powerful technique to specifically detect patterns of PSSG expression in lungs, and will enable investigations into regional changes in PSSG content in a variety of diseases.
Figures



References
-
- Shackelford RE, Heinloth AN, Heard SC, Paules RS. Cellular and molecular targets of protein S-glutathiolation. Antioxid Redox Signal. 2005;7:940–950. - PubMed
-
- Ghezzi P, Bonetto V, Fratelli M. Thiol-disulfide balance: from the concept of oxidative stress to that of redox regulation. Antioxid Redox Signal. 2005;7:964–972. - PubMed
-
- Shelton MD, Chock PB, Mieyal JJ. Glutaredoxin: role in reversible protein S-glutathionylation and regulation of redox signal transduction and protein translocation. Antioxid Redox Signal. 2005;7:348–366. - PubMed
-
- Gallogly MM, Mieyal JJ. Mechanisms of reversible protein glutathionylation in redox signaling and oxidative stress. Curr Opin Pharmacol. 2007;7:381–391. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

