Cell-free protein synthesis energized by slowly-metabolized maltodextrin
- PMID: 19558718
- PMCID: PMC2716334
- DOI: 10.1186/1472-6750-9-58
Cell-free protein synthesis energized by slowly-metabolized maltodextrin
Abstract
Background: Cell-free protein synthesis (CFPS) is a rapid and high throughput technology for obtaining proteins from their genes. The primary energy source ATP is regenerated from the secondary energy source through substrate phosphorylation in CFPS.
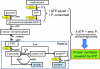
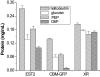
Results: Distinct from common secondary energy sources (e.g., phosphoenolpyruvate - PEP, glucose-6-phosphate), maltodextrin was used for energizing CFPS through substrate phosphorylation and the glycolytic pathway because (i) maltodextrin can be slowly catabolized by maltodextrin phosphorylase for continuous ATP regeneration, (ii) maltodextrin phosphorylation can recycle one phosphate per reaction for glucose-1-phosphate generation, and (iii) the maltodextrin chain-shortening reaction can produce one ATP per glucose equivalent more than glucose can. Three model proteins, esterase 2 from Alicyclobacillus acidocaldarius, green fluorescent protein, and xylose reductase from Neurospora crassa were synthesized for demonstration.
Conclusion: Slowly-metabolized maltodextrin as a low-cost secondary energy compound for CFPS produced higher levels of proteins than PEP, glucose, and glucose-6-phospahte. The enhancement of protein synthesis was largely attributed to better-controlled phosphate levels (recycling of inorganic phosphate) and a more homeostatic reaction environment.
Figures

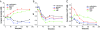
Similar articles
-
Regeneration of adenosine triphosphate from glycolytic intermediates for cell-free protein synthesis.Biotechnol Bioeng. 2001 Aug 20;74(4):309-16. Biotechnol Bioeng. 2001. PMID: 11410855
-
Energizing eukaryotic cell-free protein synthesis with glucose metabolism.FEBS Lett. 2015 Jul 8;589(15):1723-1727. doi: 10.1016/j.febslet.2015.05.045. Epub 2015 Jun 6. FEBS Lett. 2015. PMID: 26054976 Free PMC article.
-
Energy systems for ATP regeneration in cell-free protein synthesis reactions.Methods Mol Biol. 2007;375:3-17. doi: 10.1007/978-1-59745-388-2_1. Methods Mol Biol. 2007. PMID: 17634594 Review.
-
Maltose metabolism in the hyperthermophilic archaeon Thermococcus litoralis: purification and characterization of key enzymes.J Bacteriol. 1999 Jun;181(11):3358-67. doi: 10.1128/JB.181.11.3358-3367.1999. J Bacteriol. 1999. PMID: 10348846 Free PMC article.
-
The metabolic pathways of Acholeplasma and Mycoplasma: an overview.Yale J Biol Med. 1983 Sep-Dec;56(5-6):709-16. Yale J Biol Med. 1983. PMID: 6206660 Free PMC article. Review.
Cited by
-
Constructing Cell-Free Expression Systems for Low-Cost Access.ACS Synth Biol. 2022 Mar 18;11(3):1114-1128. doi: 10.1021/acssynbio.1c00342. Epub 2022 Mar 8. ACS Synth Biol. 2022. PMID: 35259873 Free PMC article.
-
A ubiquitous amino acid source for prokaryotic and eukaryotic cell-free transcription-translation systems.Front Bioeng Biotechnol. 2022 Sep 16;10:992708. doi: 10.3389/fbioe.2022.992708. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36185432 Free PMC article.
-
A Low-Cost, Thermostable, Cell-Free Protein Synthesis Platform for On-Demand Production of Conjugate Vaccines.ACS Synth Biol. 2023 Jan 20;12(1):95-107. doi: 10.1021/acssynbio.2c00392. Epub 2022 Dec 22. ACS Synth Biol. 2023. PMID: 36548479 Free PMC article.
-
A cell-free system for production of 2,3-butanediol is robust to growth-toxic compounds.Metab Eng Commun. 2019 Nov 20;10:e00114. doi: 10.1016/j.mec.2019.e00114. eCollection 2020 Jun. Metab Eng Commun. 2019. PMID: 31934547 Free PMC article.
-
Cell-free protein synthesis from non-growing, stressed Escherichia coli.Sci Rep. 2017 Nov 28;7(1):16524. doi: 10.1038/s41598-017-16767-7. Sci Rep. 2017. PMID: 29184159 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

