Regulation of matrix contraction in chronic venous disease
- PMID: 19560950
- PMCID: PMC4004365
- DOI: 10.1016/j.ejvs.2009.05.012
Regulation of matrix contraction in chronic venous disease
Abstract
Objective: The role of TGF-beta(1) in venous ulcer healing and the signalling cascades regulating dermal fibroblast function are poorly understood. To elucidate these processes, we hypothesized that TGF-beta(1) facilitates wound healing by increasing chronic venous insufficiency (CVI) induced matrix contraction via intracellular cross-talk between TGF-beta(1) and the ERK-1/2 MAP kinase signalling cascades.
Methods: Fibroblasts isolated from calf biopsies (LC) of patients with different severity of CVI (CEAP, Clinical Etiological Anatomical Pathological classes) were seeded into 200 microl collagen gels under isometric conditions. Fibroblasts from neonatal foreskins (HS68), non-CVI patients (NC), and the ipsilateral normal thigh of each CVI patient (LT) served as controls. Thirteen patients with CVI (class 2, n=5; class 4, n=5; class 6, n=3) and 2 non-CVI controls (NC, n=2) were included in the study. All experimental conditions were determined by dose-response and time-course experiments. Gels were cultured with/without 0.1 ng/ml TGF-beta(1) and with/without 50 microM PD98059 (MEK and downstream-MAPK inhibitor). Additional patient fibroblasts were transfected with constitutively active Ras (pCMV-Ras) or an empty vector (pCMV-beta) with/without 0.1 ng/ml TGF-beta(1) and with/without 50 microm PD98059. The collagen gels were released after 4 days and the percent contraction was determined by area measurements using image analysis. Differences in alpha-smooth muscle actin (alpha-SMA) and ERK-1/2 MAPK (phosphorylated and total) protein levels were analyzed with western blotting.
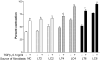
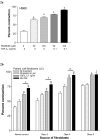
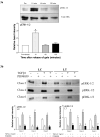
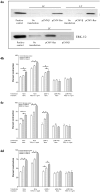
Results: Gels seeded with CVI fibroblasts contracted more than HS68, NC and LT fibroblasts. Inhibition of MAPK and/or stimulation with TGF-beta(1) increased the contraction of LC gels compared to unstimulated controls. Agonist induced gel contraction correlated with CVI disease severity. alpha-SMA protein expression in LC fibroblasts increased with MAPK inhibition with/without TGF-beta(1) stimulation, and correlated with the degree of gel contraction. Transfection with pCMV-Ras (activator of ERK-1/2) inhibited gel contraction; this inhibition was not reversed by addition of TGF-beta(1). Transfection with the pCMV-beta empty vector had no effect on gel contraction.
Conclusions: TGF-beta1 stimulation of CVI patient fibroblasts grown in 3D collagen gels results in conversion to a contractile phenotype through upregulation of alpha-SMA, and in enhanced gel contraction. Inhibition of MAPK further increases gel contraction, while Ras activation of ERK-1/2 inhibits TGF-beta1-induced gel contraction. These responses correlate with increasing CEAP severity. CVI fibroblast mediated gel contraction is therefore regulated through cross-talk between the ERK-1/2 MAPK and TGF-beta(1) signalling cascades. These data identify potentially clinically relevant therapeutic molecular targets that could enhance matrix contraction and thereby improve venous ulcer wound healing.
Conflict of interest statement
Conflict of Interest: None
Figures


Similar articles
-
TGF-β1 in Vascular Wall Pathology: Unraveling Chronic Venous Insufficiency Pathophysiology.Int J Mol Sci. 2017 Nov 26;18(12):2534. doi: 10.3390/ijms18122534. Int J Mol Sci. 2017. PMID: 29186866 Free PMC article. Review.
-
Altered proliferative responses of dermal fibroblasts to TGF-beta1 may contribute to chronic venous stasis ulcer.J Vasc Surg. 2003 Jun;37(6):1285-93. doi: 10.1016/s0741-5214(02)75295-6. J Vasc Surg. 2003. PMID: 12764277
-
Dermal tissue fibrosis in patients with chronic venous insufficiency is associated with increased transforming growth factor-beta1 gene expression and protein production.J Vasc Surg. 1999 Dec;30(6):1129-45. doi: 10.1016/s0741-5214(99)70054-6. J Vasc Surg. 1999. PMID: 10587400
-
Mitogen-activated protein kinase pathway regulates cell proliferation in venous ulcer fibroblasts.Vasc Endovascular Surg. 2006 Jan-Feb;40(1):59-66. doi: 10.1177/153857440604000108. Vasc Endovascular Surg. 2006. PMID: 16456607
-
Dermal pathology, cellular biology, and inflammation in chronic venous disease.Thromb Res. 2009;123 Suppl 4:S66-71. doi: 10.1016/S0049-3848(09)70147-1. Thromb Res. 2009. PMID: 19303508 Review.
Cited by
-
TGF-β1 in Vascular Wall Pathology: Unraveling Chronic Venous Insufficiency Pathophysiology.Int J Mol Sci. 2017 Nov 26;18(12):2534. doi: 10.3390/ijms18122534. Int J Mol Sci. 2017. PMID: 29186866 Free PMC article. Review.
-
Autogenously derived regenerative cell therapy for venous leg ulcers.Arch Med Sci Atheroscler Dis. 2018 Dec 15;3:e156-e163. doi: 10.5114/amsad.2018.81000. eCollection 2018. Arch Med Sci Atheroscler Dis. 2018. PMID: 30775606 Free PMC article.
-
Prolidase Stimulates Proliferation and Migration through Activation of the PI3K/Akt/mTOR Signaling Pathway in Human Keratinocytes.Int J Mol Sci. 2020 Dec 3;21(23):9243. doi: 10.3390/ijms21239243. Int J Mol Sci. 2020. PMID: 33287453 Free PMC article.
-
Venous endothelial injury in central nervous system diseases.BMC Med. 2013 Oct 11;11:219. doi: 10.1186/1741-7015-11-219. BMC Med. 2013. PMID: 24228622 Free PMC article. Review.
References
-
- Pappas PJ, DeFouw DO, Venezio LM, et al. Morphometric assessment of the dermal microcirculation in patients with chronic venous insufficiency. J Vasc Surg. 1997;26:784–95. - PubMed
-
- Lal BK, Saito S, Pappas PJ, et al. Altered proliferative responses of dermal fibroblasts to TGF-β1 may contribute to chronic venous stasis ulcers. J Vasc Surg. 2003;37(6):1285–93. - PubMed
-
- Pappas PJ, You R, Rameshwar P, et al. Dermal tissue fibrosis in patients with chronic venous insufficiency is associated with increased transforming growth factor-β1gene expression and protein production. J Vasc Surg. 1999;30:1129–45. - PubMed
-
- Saito S, Trovato MJ, You R, et al. Role of matrix metalloproteinases 1, 2, and 9 and tissue inhibitor of matrix metalloproteinase-1 in chronic venous insufficiency. J Vasc Surg. 2001 Nov;34(5):930–8. - PubMed
-
- Massague J. How cells read TGF-β signals. Nat Rev Mol Cell Biol. 2000;1:169–78. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

