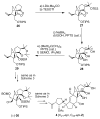
The true structures of the vannusals, part 1: Initial forays into suspected structures and intelligence gathering
- PMID: 19562816
- PMCID: PMC2777636
- DOI: 10.1002/anie.200902028
The true structures of the vannusals, part 1: Initial forays into suspected structures and intelligence gathering
Figures
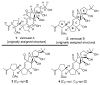

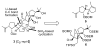
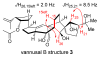
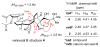
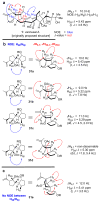
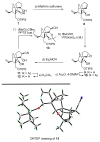
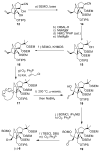
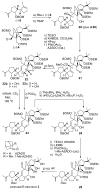
References
-
- Guella G, Callone E, Di Giuseppe G, Frassanito R, Frontini FP, Mancini I, Dini F. Eur J Org Chem. 2007:5226–5234.
-
- Nicolaou KC, et al. Angew Chem. 2009 submitted.
- Angew Chem Int Ed. 2009 submitted, see following communication in this issue.
-
-
For reviews on samarium diiodide used in organic synthesis, see: Edmonds DJ, Johnston D, Procter DJ. Chem Rev. 2004;104:3371–3403.Kagan HB. Tetrahedron. 2003;59:103510–10372.Molander GA, Harris CR. Chem Rev. 1996;96:307–308.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

