Grueneberg ganglion olfactory subsystem employs a cGMP signaling pathway
- PMID: 19565523
- PMCID: PMC4314211
- DOI: 10.1002/cne.22096
Grueneberg ganglion olfactory subsystem employs a cGMP signaling pathway
Abstract
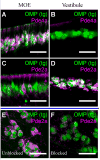
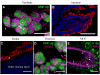
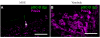
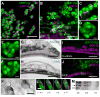
The mammalian olfactory sense employs several olfactory subsystems situated at characteristic locations in the nasal cavity to detect and report on different classes of odors. These olfactory subsystems use different neuronal signal transduction pathways, receptor expression repertoires, and axonal projection targets. The Grueneberg ganglion (GG) is a newly appreciated olfactory subsystem with receptor neurons located just inside of the nostrils that project axons to a unique domain of interconnected glomeruli in the caudal olfactory bulb. It is not well understood how the GG relates to other olfactory subsystems in contributing to the olfactory sense. Furthermore, the range of chemoreceptors and the signal transduction cascade utilized by the GG have remained mysterious. To resolve these unknowns, we explored the molecular relationship between the GG and the GC-D neurons, another olfactory subsystem that innervates similarly interconnected glomeruli in the same bulbar region. We found that mouse GG neurons express the cGMP-associated signaling proteins phosphodiesterase 2a, cGMP-dependent kinase II, and cyclic nucleotide gated channel subunit A3 coupled to a chemoreceptor repertoire of cilia-localized particulate guanylyl cyclases (pGC-G and pGC-A). The primary cGMP signaling pathway of the GG is shared with the GC-D neurons, unifying their target glomeruli as a unique center of olfactory cGMP signal transduction. However, the distinct chemoreceptor repertoire in the GG suggests that the GG is an independent olfactory subsystem. This subsystem is well suited to detect a unique set of odors and to mediate behaviors that remained intact in previous olfactory perturbations.
(c) 2009 Wiley-Liss, Inc.
Figures



Similar articles
-
The Grueneberg ganglion: signal transduction and coding in an olfactory and thermosensory organ involved in the detection of alarm pheromones and predator-secreted kairomones.Cell Tissue Res. 2021 Jan;383(1):535-548. doi: 10.1007/s00441-020-03380-w. Epub 2021 Jan 6. Cell Tissue Res. 2021. PMID: 33404842 Review.
-
Expression of cGMP signaling elements in the Grueneberg ganglion.Histochem Cell Biol. 2009 Jan;131(1):75-88. doi: 10.1007/s00418-008-0514-8. Epub 2008 Oct 1. Histochem Cell Biol. 2009. PMID: 18830617
-
Grueneberg ganglion neurons are finely tuned cold sensors.J Neurosci. 2010 Jun 2;30(22):7563-8. doi: 10.1523/JNEUROSCI.0608-10.2010. J Neurosci. 2010. PMID: 20519530 Free PMC article.
-
Odorant-evoked electrical responses in Grueneberg ganglion neurons rely on cGMP-associated signaling proteins.Neurosci Lett. 2013 Feb 28;539:38-42. doi: 10.1016/j.neulet.2013.01.032. Epub 2013 Feb 4. Neurosci Lett. 2013. PMID: 23384572
-
The Grueneberg ganglion: a novel sensory system in the nose.Histol Histopathol. 2010 Jul;25(7):909-15. doi: 10.14670/HH-25.909. Histol Histopathol. 2010. PMID: 20503179 Review.
Cited by
-
The cyclic nucleotide-gated ion channel CNGA3 contributes to coolness-induced responses of Grueneberg ganglion neurons.Cell Mol Life Sci. 2010 Jun;67(11):1859-69. doi: 10.1007/s00018-010-0296-8. Epub 2010 Feb 18. Cell Mol Life Sci. 2010. PMID: 20165899 Free PMC article.
-
P/Q Type Calcium Channel Cav2.1 Defines a Unique Subset of Glomeruli in the Mouse Olfactory Bulb.Front Cell Neurosci. 2018 Sep 4;12:295. doi: 10.3389/fncel.2018.00295. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30233329 Free PMC article.
-
The Grueneberg ganglion: signal transduction and coding in an olfactory and thermosensory organ involved in the detection of alarm pheromones and predator-secreted kairomones.Cell Tissue Res. 2021 Jan;383(1):535-548. doi: 10.1007/s00441-020-03380-w. Epub 2021 Jan 6. Cell Tissue Res. 2021. PMID: 33404842 Review.
-
Innate Predator Odor Aversion Driven by Parallel Olfactory Subsystems that Converge in the Ventromedial Hypothalamus.Curr Biol. 2015 May 18;25(10):1340-6. doi: 10.1016/j.cub.2015.03.026. Epub 2015 Apr 30. Curr Biol. 2015. PMID: 25936549 Free PMC article.
-
Neural mechanisms of alarm pheromone signaling.Mol Cells. 2013 Mar;35(3):177-81. doi: 10.1007/s10059-013-0056-3. Epub 2013 Mar 5. Mol Cells. 2013. PMID: 23471444 Free PMC article. Review.
References
-
- Bender AT, Beavo JA. Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol Rev. 2006;58(3):488–520. - PubMed
-
- Biel M, Michalakis S. Function and dysfunction of CNG channels: insights from channelopathies and mouse models. Mol Neurobiol. 2007;35(3):266–277. - PubMed
-
- Bolger GB, McPhee I, Houslay MD. Alternative splicing of cAMP-specific phosphodiesterase mRNA transcripts. Characterization of a novel tissue-specific isoform, RNPDE4A8. J Biol Chem. 1996;271(2):1065–1071. - PubMed
-
- Brechbuhl J, Klaey M, Broillet MC. Grueneberg ganglion cells mediate alarm pheromone detection in mice. Science. 2008;321(5892):1092–1095. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

