Human cortical prostheses: lost in translation?
- PMID: 19569893
- PMCID: PMC3614414
- DOI: 10.3171/2009.4.FOCUS0987
Human cortical prostheses: lost in translation?
Abstract
Direct brain control of a prosthetic system is the subject of much popular and scientific news. Neural technology and science have advanced to the point that proof-of-concept systems exist for cortically-controlled prostheses in rats, monkeys, and even humans. However, realizing the dream of making such technology available to everyone is still far off. Fortunately today there is great public and scientific interest in making this happen, but it will only occur when the functional benefits of such systems outweigh the risks. In this article, the authors briefly summarize the state of the art and then highlight many issues that will directly limit clinical translation, including system durability, system performance, and patient risk. Despite the challenges, scientists and clinicians are in the desirable position of having both public and fiscal support to begin addressing these issues directly. The ultimate challenge now is to determine definitively whether these prosthetic systems will become clinical reality or forever unrealized.
Figures
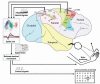


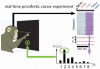


References
-
- Achtman N, Afshar A, Santhanam G, Yu B, Ryu SI, Shenoy KV. Free-paced high performance brain-computer interfaces. J Neural Eng. 2007;4:336–347. - PubMed
-
- Adelson PD, Black PM, Madsen JR, Kramer U, Rockoff MA, Riviello JJ, et al. Use of subdural grids and strip electrodes to identify a seizure focus in children. Pediatr Neurosurg. 1995;22:174–180. - PubMed
-
- Badran K, Arya AK, Bunstone D, Mackinnon N. Long-term complications of bone-anchored hearing aids: a 14-year experience. J Laryngol Otol. 2009;123:170–176. - PubMed
-
- Bauman JA, Feoli E, Romanelli P, Doyle WK, Devinsky O, Weiner HL. Multistage epilepsy surgery: safety, efficacy, and utility of a novel approach in pediatric extratemporal epilepsy. Neurosurgery. 2005;56:318–334. - PubMed
-
- Birbaumer N, Ghanayim N, Hinterberger T, Iversen I, Kotchoubey B, Kubler A, et al. A spelling device for the paralysed. Nature. 1999;398:297–298. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

