Getting signals into the brain: visual prosthetics through thalamic microstimulation
- PMID: 19569894
- PMCID: PMC2848996
- DOI: 10.3171/2009.4.FOCUS0986
Getting signals into the brain: visual prosthetics through thalamic microstimulation
Abstract
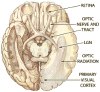
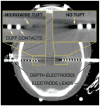
Common causes of blindness are diseases that affect the ocular structures, such as glaucoma, retinitis pigmentosa, and macular degeneration, rendering the eyes no longer sensitive to light. The visual pathway, however, as a predominantly central structure, is largely spared in these cases. It is thus widely thought that a device-based prosthetic approach to restoration of visual function will be effective and will enjoy similar success as cochlear implants have for restoration of auditory function. In this article the authors review the potential locations for stimulation electrode placement for visual prostheses, assessing the anatomical and functional advantages and disadvantages of each. Of particular interest to the neurosurgical community is placement of deep brain stimulating electrodes in thalamic structures that has shown substantial promise in an animal model. The theory of operation of visual prostheses is discussed, along with a review of the current state of knowledge. Finally, the visual prosthesis is proposed as a model for a general high-fidelity machine-brain interface.
Conflict of interest statement
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Figures

References
-
- Bak M, Girvin JP, Hambrecht FT, Kufta CV, Loeb GE, Schmidt EM. Visual sensations produced by intracortical microstimulation of the human occipital cortex. Med Biol Eng Comput. 1990;28:257–259. - PubMed
-
- Bradley DC, Troyk PR, Berg JA, Bak M, Cogan S, Erickson R, et al. Visuotopic mapping through a multichannel stimulating implant in primate V1. J Neurophysiol. 2005;93:1659–1670. - PubMed
-
- Brelén ME, De Potter P, Gersdorff M, Cosnard G, Veraart C, Delbeke J. Intraorbital implantation of a stimulating electrode for an optic nerve visual prosthesis. J Neurosurg. 2006;104:593–597. - PubMed
-
- Brindley GS. Physiology of the Retina and the Visual Pathway. London: Edward Arnold Ltd; 1960.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

