Structural and synthetic investigations of tanikolide dimer, a SIRT2 selective inhibitor, and tanikolide seco-acid from the Madagascar marine cyanobacterium Lyngbya majuscula
- PMID: 19572575
- PMCID: PMC2736347
- DOI: 10.1021/jo900578j
Structural and synthetic investigations of tanikolide dimer, a SIRT2 selective inhibitor, and tanikolide seco-acid from the Madagascar marine cyanobacterium Lyngbya majuscula
Abstract
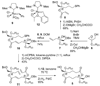
Tanikolide seco-acid 2 and tanikolide dimer 3, the latter a novel and selective SIRT2 inhibitor, were isolated from the Madagascar marine cyanobacterium Lyngbya majuscula. The structure of 2, isolated as the pure R enantiomer, was elucidated by X-ray experiment in conjunction with NMR and optical rotation data, whereas the depside molecular structure of 3 was initially thought to be a meso compound as established by NMR, MS, and chiral HPLC analyses. Subsequent total synthesis of the three tanikolide dimer stereoisomers 4, 5, and ent-5, followed by chiral GC-MS comparisons with the natural product, showed it to be exclusively the R,R-isomer 5. Tanikolide dimer 3 (= 5) inhibited SIRT2 with an IC(50) = 176 nM in one assay format and 2.4 microM in another. Stereochemical determination of symmetrical dimers such as compound 3 pose intriguing and subtle questions in structure elucidation and, as shown in the current work, are perhaps best answered in conjunction with total synthesis.
Figures
Similar articles
-
Malyngolide dimer, a bioactive symmetric cyclodepside from the panamanian marine cyanobacterium Lyngbya majuscula.J Nat Prod. 2010 Apr 23;73(4):709-11. doi: 10.1021/np9005184. J Nat Prod. 2010. PMID: 20158242 Free PMC article.
-
Tanikolide, a toxic and antifungal lactone from the marine cyanobacterium Lyngbya majuscula.J Nat Prod. 1999 Sep;62(9):1333-5. doi: 10.1021/np990162c. J Nat Prod. 1999. PMID: 10514329
-
Curacin D, an antimitotic agent from the marine cyanobacterium Lyngbya majuscula.Phytochemistry. 1998 Dec;49(8):2387-9. doi: 10.1016/s0031-9422(98)00365-3. Phytochemistry. 1998. PMID: 9887531
-
Caylobolide A, a unique 36-membered macrolactone from a Bahamian Lyngbya majuscula.Org Lett. 2002 May 2;4(9):1535-8. doi: 10.1021/ol025759p. Org Lett. 2002. PMID: 11975622
-
Selective SIRT2 inhibitors as promising anticancer therapeutics: An update from 2016 to 2020.Eur J Med Chem. 2021 Nov 15;224:113709. doi: 10.1016/j.ejmech.2021.113709. Epub 2021 Jul 18. Eur J Med Chem. 2021. PMID: 34303869 Review.
Cited by
-
Sirtuin modulators.Handb Exp Pharmacol. 2011;206:241-55. doi: 10.1007/978-3-642-21631-2_11. Handb Exp Pharmacol. 2011. PMID: 21879453 Free PMC article. Review.
-
Influence of Low Salt Concentration on Growth Behavior and General Biomass Composition in Lyngbya purpurem (Cyanobacteria).Mar Drugs. 2020 Dec 4;18(12):621. doi: 10.3390/md18120621. Mar Drugs. 2020. PMID: 33291783 Free PMC article.
-
Biological targets and mechanisms of action of natural products from marine cyanobacteria.Nat Prod Rep. 2015 Mar;32(3):478-503. doi: 10.1039/c4np00104d. Nat Prod Rep. 2015. PMID: 25571978 Free PMC article. Review.
-
Marine Microorganism-Derived Macrolactins Inhibit Inflammatory Mediator Effects in LPS-Induced Macrophage and Microglial Cells by Regulating BACH1 and HO-1/Nrf2 Signals through Inhibition of TLR4 Activation.Molecules. 2020 Feb 4;25(3):656. doi: 10.3390/molecules25030656. Molecules. 2020. PMID: 32033079 Free PMC article.
-
Zebrafish-based identification of the antiseizure nucleoside inosine from the marine diatom Skeletonema marinoi.PLoS One. 2018 Apr 24;13(4):e0196195. doi: 10.1371/journal.pone.0196195. eCollection 2018. PLoS One. 2018. PMID: 29689077 Free PMC article.
References
-
- Singh IP, Milligan KE, Gerwick WH. J. Nat. Prod. 1999;62:1333–1335. - PubMed
-
- Vichare P, Chattopadhyay A. Tetrahedron: Asymmetry. 2008;19:598–602.
- Zhang C, Hosoda N, Asami M. Tetrahedron: Asymmetry. 2007;18:2185–2189.
- Fujioka Hiromichi, Matsuda Satoshi, Horai Mai, Fujii Eri, Morishita Maiko, Nishiguchi Natsuko, Hata Kayoko, Kita Yasuyuki. Chem. --Eur. J. 2007;13:5238–5248. - PubMed
- Wu Fanghui, Hong Ran, Khan Jihan, Liu Xiaofeng, Deng Li. Angew. Chem., Int. Ed. 2006;45:4301–4305. - PubMed
- Kita Y, Matsuda S, Fujii E, Horai M, Hata K, Fujioka H. Angew. Chem., Int. Ed. 2005;44:5857–5860. - PubMed
- Yajima T, Saito C, Nagano H. Tetrahedron. 2005;61:10203–10215.
- Ohgiya T, Nakamura K, Nishiyama S. Bull. Chem. Soc. Jpn. 2005;78:1549–1554.
- Ohgiya T, Nishiyama S. Tetrahedron Lett. 2004;45:8273–8275.
- Arasaki H, Iwata M, Makida M, Masaki Y. Chem. Pharm. Bull. 2004;52:848–852. - PubMed
- Schomaker JM, Borhan B. Org. Biomol. Chem. 2004;2:621–624. - PubMed
- Koumbis AE, Dieti KM, Vikentiou MG, Gallos JK. Tetrahedron Lett. 2003;44:2513–2516.
- Carda M, Rodriguez S, Castillo E, Bellido A, Diaz-Oltra S, Alberto MJ. Tetrahedron. 2003;59:857–864.
- Mizutani H, Watanabe M, Honda T. Tetrahedron. 2002;58:8929–8936.
- Tanaka H, Kozuki Y, Ogasawara K. Tetrahedron Lett. 2002;43:4175–4178.
- Kanada MR, Taniguchi T, Ogasawara K. Synlett. 2000;7:1019–1021.
-
- Andrianasolo EH, Gross H, Goeger D, Musafija-Girt M, McPhail K, Leal RM, Mooberry SL, Gerwick WH. Org. Lett. 2005;7:1375–1378. - PubMed
-
- Han B, Goeger D, Maier CS, Gerwick WH. J. Org. Chem. 2005;70:3133–3139. - PubMed
-
- Crews P, Gerwick W, Schmitz F, France D, Bair K, Wright A, Hallock Y. Pharmaceutical Biology (Lisse, Netherlands) 2005;1 Suppl. 1:39–52.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous