Virus binding to a plasma membrane receptor triggers interleukin-1 alpha-mediated proinflammatory macrophage response in vivo
- PMID: 19576795
- PMCID: PMC2759279
- DOI: 10.1016/j.immuni.2009.04.015
Virus binding to a plasma membrane receptor triggers interleukin-1 alpha-mediated proinflammatory macrophage response in vivo
Abstract
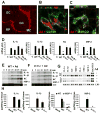
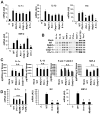
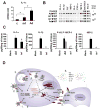
The recognition of viral components by host pattern-recognition receptors triggers the induction of the antiviral innate immune response. Toll-like receptor 9 (TLR9) and NLRP3 inflammasome were shown to be the principal specific sensors of viral double-stranded DNA. Here we present evidence that macrophages in vivo activated an innate immune response to a double-stranded DNA virus, adenovirus (Ad), independently of TLR9 or NLRP3 inflammasome. In response to Ad, macrophage-derived IL-1 alpha triggered IL-1RI-dependent production of a defined set of proinflammatory cytokines and chemokines. The IL-1 alpha-mediated response required a selective interaction of virus arginine-glycine-aspartic acid (RGD) motifs with macrophage beta(3) integrins. Thus, these data identify IL-1 alpha-IL-1RI as a key pathway allowing for the activation of proinflammatory responses to the virus, independently of its genomic nucleic acid recognition.
Figures




Comment in
-
Integr-ating IL-1 alpha in antiviral host defenses.Immunity. 2009 Jul 17;31(1):7-9. doi: 10.1016/j.immuni.2009.07.001. Immunity. 2009. PMID: 19604487
References
-
- Attur MG, Dave MN, Clancy RR, Patel IR, Abramson SB, Amin AR. Functional genomic analysis in arthritis-affected cartilage: Yin-yang regulation of inflammatory mediators by alpha(5)beta(1) and alpha(v)beta(3) integrins. Journal of Immunology. 2000;164:2684–2691. - PubMed
-
- Brunetti-Pierri N, Palmer DJ, Beaudet AL, Carey KD, Finegold M, Ng P. Acute toxicity after high-dose systemic injection of helper-dependent adenoviral vectors into nonhuman primates. Hum Gene Ther. 2004;15:35–46. - PubMed
-
- Chang CH, Huang Y, Anderson R. Activation of vascular endothelial cells by IL-1 alpha released by epithelial cells infected with respiratory syncytial virus. Cellular Immunology. 2003;221:37–41. - PubMed
-
- Chen CJ, Kono H, Golenbock D, Reed G, Akira S, Rock KL. Identification of a key pathway required for the sterile inflammatory response triggered by dying cells. Nature Medicine. 2007;13:851–856. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

