Adenylate cyclase regulates elongation of mammalian primary cilia
- PMID: 19576885
- PMCID: PMC3161028
- DOI: 10.1016/j.yexcr.2009.06.028
Adenylate cyclase regulates elongation of mammalian primary cilia
Abstract
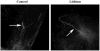
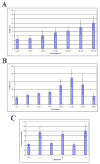
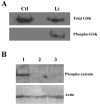
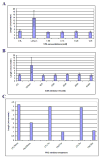
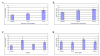
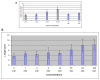
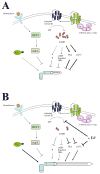
The primary cilium is a non-motile microtubule-based structure that shares many similarities with the structures of flagella and motile cilia. It is well known that the length of flagella is under stringent control, but it is not known whether this is true for primary cilia. In this study, we found that the length of primary cilia in fibroblast-like synoviocytes, either in log phase culture or in quiescent state, was confined within a range. However, when lithium was added to the culture to a final concentration of 100 mM, primary cilia of synoviocytes grew beyond this range, elongating to a length that was on average approximately 3 times the length of untreated cilia. Lithium is a drug approved for treating bipolar disorder. We dissected the molecular targets of this drug, and observed that inhibition of adenylate cyclase III (ACIII) by specific inhibitors mimicked the effects of lithium on primary cilium elongation. Inhibition of GSK-3beta by four different inhibitors did not induce primary cilia elongation. ACIII was found in primary cilia of a variety of cell types, and lithium treatment of these cell types led to their cilium elongation. Further, we demonstrate that different cell types displayed distinct sensitivities to the lithium treatment. However, in all cases examined primary cilia elongated as a result of lithium treatment. In particular, two neuronal cell types, rat PC-12 adrenal medulla cells and human astrocytes, developed long primary cilia when lithium was used at or close to the therapeutic relevant concentration (1-2 mM). These results suggest that the length of primary cilia is controlled, at least in part, by the ACIII-cAMP signaling pathway.
Figures


References
-
- Alieva IB, Vorobjev IA. Vertebrate primary cilia: a sensory part of centrosomal complex in tissue cells, but a “sleeping beauty” in cultured cells? Cell Biol Int. 2004;28:139–150. - PubMed
-
- Allison JH, Stewart MA. Reduced brain inositol in lithium-treated rats. Nat New Biol. 1971;233:267–268. - PubMed
-
- Badano JL, Mitsuma N, Beales PL, Katsanis N. The ciliopathies: an emerging class of human genetic disorders. Annu Rev Genomics Hum Genet. 2006;7:125–148. - PubMed
-
- Bakalyar HA, Reed RR. Identification of a specialized adenylyl cyclase that may mediate odorant detection. Science. 1990;250:1403–1406. - PubMed
-
- Berbari NF, Bishop GA, Askwith CC, Lewis JS, Mykytyn K. Hippocampal neurons possess primary cilia in culture. J Neurosci Res. 2007;85:1095–1100. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

