The feasibility of a single-blinded fast-track pragmatic randomised controlled trial of a complex intervention for breathlessness in advanced disease
- PMID: 19583857
- PMCID: PMC2731082
- DOI: 10.1186/1472-684X-8-9
The feasibility of a single-blinded fast-track pragmatic randomised controlled trial of a complex intervention for breathlessness in advanced disease
Abstract
Background: The Breathlessness Intervention Service is a novel service for patients with intractable breathlessness regardless of aetiology. It is being evaluated using the Medical Research Council's framework for the evaluation of complex interventions. This paper describes the feasibility results of Phase II: a single-blinded fast-track pragmatic randomised controlled trial.
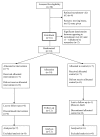
Methods: A single-blinded fast-track pragmatic randomised controlled trial was conducted for patients with chronic obstructive pulmonary disease referred to the service. Patients were randomised to either receive the intervention immediately for an eight-week period, or receive the intervention after an eight-week period on a waiting list during which time they received standard care. Outcomes examined included: response rates to the trial; response rates to the individual questionnaires and items; comments relating to the trial functioning made during interviews with patients, carers, referrers and service providers; and, researcher fieldwork notes.
Results: 16 of the 20 eligible patients agreed to participate in a recruitment visit (16/20); 14 respondents went on to complete a recruitment visit/baseline interview. The majority of those who completed a recruitment visit/baseline interview completed the RCT protocol (13/14); 12 of their carers were recruited and completed the protocol. An unblinding rate of 6/25 respondents (patients and carers) was identified. Missing data were minimal and only one patient was lost to follow up. The fast-track trial methodology proved feasible and acceptable. Two of the baseline/outcome measures proved unsuitable: the WHO performance scale and the Schedule for the Evaluation of Individual Quality of Life-Direct Weighting (SEIQoL-DW).
Conclusion: This study adds to the evidence that fast-track randomised controlled trials are feasible and acceptable in evaluations of palliative care interventions for patients with non-malignant conditions. Reasonable response rates and low attrition rates were achieved. Further, with adequate preparation of the research and randomisation teams, clinicians, and responders, and effective liaison with the clinicians, single-blinding proved possible. Methods were identified to reduce unblinding through careful attention to the type of data collected at unblinded measurement points; the content of interviews should be carefully considered when designing blinded-trial protocols.
Trial registration: Clinical Trials.gov NCT00711438.
Figures
References
Associated data
LinkOut - more resources
Full Text Sources
Medical