Using social networks to recruit an HIV vaccine preparedness cohort
- PMID: 19584741
- PMCID: PMC2783777
- DOI: 10.1097/QAI.0b013e3181acff91
Using social networks to recruit an HIV vaccine preparedness cohort
Abstract
Objectives: To evaluate a social network approach to develop an adolescent cohort for HIV vaccine preparedness and investigate characteristics that influence recruitment.
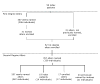
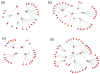
Methods: We summarize baseline data from a prospective cohort study that included 4 sessions over 6 months. Fifty-nine HIV-infected adolescent and adult patients of a family-based HIV clinic named significant others and indicated willingness to involve them in this study. Sixty-two adolescent and adult significant others not known to be HIV infected were enrolled. Logistic regression was used to estimate factors associated with willingness.
Results: Participants identified 624 social network members including 276 adolescents (44%). Network member's awareness of the index's HIV positivity (P < 0.01) and older age (P = 0.05) affected willingness. Respondents were less willing to invite drug-risk alters (P = 0.006). Adolescents were willing to invite more adolescents than were adults (P < 0.0001). Adolescents younger than 18 years old reported fewer sexual and drug-using risk behaviors than expected.
Conclusions: HIV-infected patients are willing to recruit their social networks, provided concerns about disclosure of HIV status are addressed. Using social networks to identify and recruit adolescent populations is appropriate and feasible for vaccine preparedness activities, future vaccine trials, and other prevention programs, but procedures are needed to selectively identify and retain high-risk youth.
Figures
Similar articles
-
Young at risk-people in Maputo City, Mozambique, present a high willingness to participate in HIV trials: Results from an HIV vaccine preparedness cohort study.PLoS One. 2021 Dec 2;16(12):e0260126. doi: 10.1371/journal.pone.0260126. eCollection 2021. PLoS One. 2021. PMID: 34855790 Free PMC article.
-
Contextualizing willingness to participate: recommendations for engagement, recruitment & enrolment of Kenyan MSM in future HIV prevention trials.BMC Public Health. 2017 May 18;17(1):469. doi: 10.1186/s12889-017-4395-4. BMC Public Health. 2017. PMID: 28521748 Free PMC article.
-
Recruitment using respondent driven sampling, risk behaviors assessment and willingness of young female sex workers (18-25 years) in Dar Es Salaam, Tanzania to participate in HIV vaccine trials.BMC Public Health. 2019 Nov 20;19(1):1537. doi: 10.1186/s12889-019-7822-x. BMC Public Health. 2019. PMID: 31796002 Free PMC article.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Challenges to conducting HIV preventative vaccine trials with adolescents.J Acquir Immune Defic Syndr. 2004 Jun 1;36(2):726-33. doi: 10.1097/00126334-200406010-00010. J Acquir Immune Defic Syndr. 2004. PMID: 15167292 Review.
Cited by
-
Hepatitis C vaccine clinical trials among people who use drugs: potential for participation and involvement in recruitment.Contemp Clin Trials. 2015 Mar;41:9-16. doi: 10.1016/j.cct.2014.12.015. Epub 2014 Dec 30. Contemp Clin Trials. 2015. PMID: 25553715 Free PMC article.
-
Vaccine research, adolescents and Africa.Hum Vaccin Immunother. 2013 Jan;9(1):108-11. doi: 10.4161/hv.22143. Epub 2012 Oct 29. Hum Vaccin Immunother. 2013. PMID: 23108358 Free PMC article. No abstract available.
-
Protocol for a mixed-methods feasibility study for the surviving opioid overdose with naloxone education and resuscitation (SOONER) randomised control trial.BMJ Open. 2019 Nov 12;9(11):e029436. doi: 10.1136/bmjopen-2019-029436. BMJ Open. 2019. PMID: 31722937 Free PMC article.
-
Barriers to adolescents' participation in HIV biomedical prevention research.J Acquir Immune Defic Syndr. 2010 Jul;54 Suppl 1(Suppl 1):S12-7. doi: 10.1097/QAI.0b013e3181e1e2c0. J Acquir Immune Defic Syndr. 2010. PMID: 20571418 Free PMC article.
-
SRSF3 and SRSF7 modulate 3'UTR length through suppression or activation of proximal polyadenylation sites and regulation of CFIm levels.Genome Biol. 2021 Mar 11;22(1):82. doi: 10.1186/s13059-021-02298-y. Genome Biol. 2021. PMID: 33706811 Free PMC article.
References
-
- Coletti AS, Heagerty P, Sheon AR, et al. Randomized, controlled evaluation of a prototype informed consent process for HIV vaccine efficacy trials. J Acquir Immune Defic Syndr. 2003;32(2):161–9. - PubMed
-
- Buchbinder SP, Metch B, Holte SE, et al. Determinants of enrollment in a preventive HIV vaccine trial: Hypothetical versus actual willingness and barriers to participation. J Acquir Immune Defic Syndr. 2004;36(1):604–12. - PubMed
-
- Bekker LG, Jaspan H, McIntyre J, et al. Adolescents and HIV vaccine trials: What are the clinical trial site issues? J Int Assoc Physicians AIDS Care. 2005;4(4):93–7. - PubMed
-
- Stanford PD, Monte DA, Briggs FM, et al. Recruitment and Retention of Adolescent Participants in HIV Research: Findings from the REACH (Reaching for Excellence in Adolescent Care and Health) Project. J Adolesc Health. 2003;32(3):192–203. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

