Dlg1, Sec8, and Mtmr2 regulate membrane homeostasis in Schwann cell myelination
- PMID: 19587293
- PMCID: PMC6664895
- DOI: 10.1523/JNEUROSCI.1423-09.2009
Dlg1, Sec8, and Mtmr2 regulate membrane homeostasis in Schwann cell myelination
Abstract
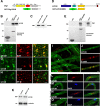
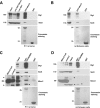
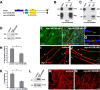
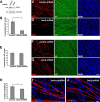
How membrane biosynthesis and homeostasis is achieved in myelinating glia is mostly unknown. We previously reported that loss of myotubularin-related protein 2 (MTMR2) provokes autosomal recessive demyelinating Charcot-Marie-Tooth type 4B1 neuropathy, characterized by excessive redundant myelin, also known as myelin outfoldings. We generated a Mtmr2-null mouse that models the human neuropathy. We also found that, in Schwann cells, Mtmr2 interacts with Discs large 1 (Dlg1), a scaffold involved in polarized trafficking and membrane addition, whose localization in Mtmr2-null nerves is altered. We here report that, in Schwann cells, Dlg1 also interacts with kinesin 13B (kif13B) and Sec8, which are involved in vesicle transport and membrane tethering in polarized cells, respectively. Taking advantage of the Mtmr2-null mouse as a model of impaired membrane formation, we provide here the first evidence for a machinery that titrates membrane formation during myelination. We established Schwann cell/DRG neuron cocultures from Mtmr2-null mice, in which myelin outfoldings were reproduced and almost completely rescued by Mtmr2 replacement. By exploiting this in vitro model, we propose a mechanism whereby kif13B kinesin transports Dlg1 to sites of membrane remodeling where it coordinates a homeostatic control of myelination. The interaction of Dlg1 with the Sec8 exocyst component promotes membrane addition, whereas with Mtmr2, negatively regulates membrane formation. Myelin outfoldings thus arise as a consequence of the loss of negative control on the amount of membrane, which is produced during myelination.
Figures





References
-
- Anitei M, Ifrim M, Ewart MA, Cowan AE, Carson JH, Bansal R, Pfeiffer SE. A role for Sec8 in oligodendrocyte morphological differentiation. J Cell Sci. 2006;119:807–818. - PubMed
-
- Asaba N, Hanada T, Takeuchi A, Chishti AH. Direct interaction with a kinesin-related motor mediates transport of mammalian discs large tumor suppressor homologue in epithelial cells. J Biol Chem. 2003;278:8395–8400. - PubMed
-
- Bolino A, Muglia M, Conforti FL, LeGuern E, Salih MA, Georgiou DM, Christodoulou K, Hausmanowa-Petrusewicz I, Mandich P, Schenone A, Gambardella A, Bono F, Quattrone A, Devoto M, Monaco AP. Charcot-Marie-Tooth type 4B is caused by mutations in the gene encoding myotubularin-related protein-2. Nat Genet. 2000;25:17–19. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
