Mannitol-facilitated CNS entry of rAAV2 vector significantly delayed the neurological disease progression in MPS IIIB mice
- PMID: 19587708
- PMCID: PMC4289609
- DOI: 10.1038/gt.2009.85
Mannitol-facilitated CNS entry of rAAV2 vector significantly delayed the neurological disease progression in MPS IIIB mice
Abstract
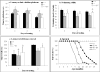
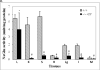
The presence of the blood-brain barrier (BBB) presents the most critical challenge in therapeutic development for mucopolysaccharidosis (MPS) IIIB, a lysosomal storage disease with severe neurological manifestation, because of alpha-N-acetylglucosaminidase (NaGlu) deficiency. Earlier, we showed a global central nervous system (CNS) transduction in mice by mannitol-facilitated entry of intravenous (IV)-delivered recombinant adeno-associated viral serotype 2 (rAAV2) vector. In this study, we optimized the approach and showed that the maximal transduction in the CNS occurred when the rAAV2 vector was IV injected at 8 min after mannitol administration, and was approximately 10-fold more efficient than IV delivery of the vector at 5 or 10 min after mannitol infusion. Using this optimal (8 min) regimen, a single IV infusion of rAAV2-CMV-hNaGlu vector is therapeutically beneficial for treating the CNS disease of MPS IIIB in adult mice, with significantly extended survival, improved behavioral performance, and reduction of brain lysosomal storage pathology. The therapeutic benefit correlated with maximal delivery to the CNS, but not peripheral tissues. This milestone data shows the first effective gene delivery across the BBB to treat CNS disease. The critical timing of vector delivery and mannitol infusion highlights the important contribution of this pretreatment to successful intervention, and the long history of safe use of mannitol in patients bodes well for its application in CNS gene therapy.
Figures









References
-
- Neufeld EF, Muenzer J. The mucopolysaccharidoses. In: Scriver CR, Beaudet AL, Sly WS, Valle D, editors. The metabolic & molecular basis of inherited disease. 8. McGraw-Hill; New York, St Louis; San Francisco: 2001. pp. 3421–3452.
-
- McGlynn R, Dobrenis K, Walkley SU. Differential subcellular localization of cholesterol, gangliosides, and glycosaminoglycans in murine models of mucopolysaccharide storage disorders. J Comp Neurol. 2004;480:415–426. - PubMed
-
- Villani GR, Gargiulo N, Faraonio R, Castaldo S, Gonzalez YRE, Di Natale P. Cytokines, neurotrophins, and oxidative stress in brain disease from mucopolysaccharidosis IIIB. J Neurosci Res. 2007;85:612–622. - PubMed
-
- Dirosario J, Divers E, Wang C, Etter J, Charrier A, Jukkola P, et al. Innate and adaptive immune activation in the brain of MPS IIIB mouse model. J Neurosci Res. 2008 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

