Alterations in peroxisome proliferator-activated receptor mRNA expression in skeletal muscle after acute and repeated bouts of exercise
- PMID: 19588229
- PMCID: PMC2801580
- DOI: 10.1007/s11010-009-0195-1
Alterations in peroxisome proliferator-activated receptor mRNA expression in skeletal muscle after acute and repeated bouts of exercise
Abstract
Peroxisome proliferator-activated receptors (PPAR) exist in three different forms, alpha (alpha), beta/delta (beta/delta), or gamma (gamma), all of which are expressed in skeletal muscle and play a critical role in the regulation of oxidative metabolism. The purpose of this investigation was to determine the mRNA expression pattern of the different PPARs and peroxisome proliferator-activated receptor alpha coactivator-1 alpha (PGC-1alpha) in muscles that largely rely on either glycolytic (plantaris) or oxidative (soleus) metabolism. Further, we also examined the alterations in the PPARs mRNA expression after one bout of endurance exercise or after 12 weeks of exercise training in the different muscles. Female Sprague-Dawley rats (5-8 months) were either run on the treadmill once or exercised trained for 12 weeks. The muscles were removed 24 h after the last bout of exercise. The results demonstrated with the exception of PPAR beta/delta, the PPAR mRNAs are expressed to a greater extent in the soleus muscle than in the plantaris muscle in sedentary animals. PPARgamma was the least abundantly expressed PPAR in either the soleus or the plantaris muscle. With respect to exercise training, only PPARgamma mRNA expression increased in the soleus muscle, while PPARbeta/delta and gamma mRNA levels increased in the plantaris muscle. Minimal changes were detected in any of the PPARs with one bout of exercise training. These results suggest that PPARgamma mRNA levels are the lowest in skeletal muscle among all of the PPARs and PPARgamma mRNA is the most responsive to changes in physical activity levels.
Figures
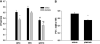
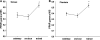

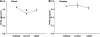
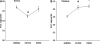

Similar articles
-
Thyroid hormone activation by type 2 deiodinase mediates exercise-induced peroxisome proliferator-activated receptor-γ coactivator-1α expression in skeletal muscle.J Physiol. 2016 Sep 15;594(18):5255-69. doi: 10.1113/JP272440. Epub 2016 Aug 18. J Physiol. 2016. PMID: 27302464 Free PMC article.
-
An increase in murine skeletal muscle peroxisome proliferator-activated receptor-gamma coactivator-1alpha (PGC-1alpha) mRNA in response to exercise is mediated by beta-adrenergic receptor activation.Endocrinology. 2007 Jul;148(7):3441-8. doi: 10.1210/en.2006-1646. Epub 2007 Apr 19. Endocrinology. 2007. PMID: 17446185
-
Endurance exercise increases the SIRT1 and peroxisome proliferator-activated receptor gamma coactivator-1alpha protein expressions in rat skeletal muscle.Metabolism. 2008 Jul;57(7):986-98. doi: 10.1016/j.metabol.2008.02.017. Metabolism. 2008. PMID: 18555842
-
PGC-1alpha is not mandatory for exercise- and training-induced adaptive gene responses in mouse skeletal muscle.Am J Physiol Endocrinol Metab. 2008 Feb;294(2):E463-74. doi: 10.1152/ajpendo.00666.2007. Epub 2007 Dec 11. Am J Physiol Endocrinol Metab. 2008. PMID: 18073319
-
The Regulatory Roles of PPARs in Skeletal Muscle Fuel Metabolism and Inflammation: Impact of PPAR Agonism on Muscle in Chronic Disease, Contraction and Sepsis.Int J Mol Sci. 2021 Sep 10;22(18):9775. doi: 10.3390/ijms22189775. Int J Mol Sci. 2021. PMID: 34575939 Free PMC article. Review.
Cited by
-
Transcriptional modulation of mitochondria biogenesis pathway at and above critical speed in mice.Mol Cell Biochem. 2015 Jul;405(1-2):223-32. doi: 10.1007/s11010-015-2413-3. Epub 2015 Apr 26. Mol Cell Biochem. 2015. PMID: 25912548
-
Activation of PPARδ signaling improves skeletal muscle oxidative metabolism and endurance function in an animal model of ischemic left ventricular dysfunction.Am J Physiol Heart Circ Physiol. 2015 May 1;308(9):H1078-85. doi: 10.1152/ajpheart.00679.2014. Epub 2015 Feb 20. Am J Physiol Heart Circ Physiol. 2015. PMID: 25713305 Free PMC article.
-
Women have higher protein content of beta-oxidation enzymes in skeletal muscle than men.PLoS One. 2010 Aug 6;5(8):e12025. doi: 10.1371/journal.pone.0012025. PLoS One. 2010. PMID: 20700461 Free PMC article.
-
PPARγ regulates inflammatory reaction by inhibiting the MAPK/NF-κB pathway in C2C12 skeletal muscle cells.J Physiol Biochem. 2017 Feb;73(1):49-57. doi: 10.1007/s13105-016-0523-3. Epub 2016 Oct 7. J Physiol Biochem. 2017. PMID: 27718123
-
Exercise-induced BDNF promotes PPARδ-dependent reprogramming of lipid metabolism in skeletal muscle during exercise recovery.Sci Signal. 2024 Mar 19;17(828):eadh2783. doi: 10.1126/scisignal.adh2783. Epub 2024 Mar 19. Sci Signal. 2024. PMID: 38502732 Free PMC article.
References
-
- Holloszy JO. Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J Biol Chem. 1967;242:2278–2282. - PubMed
-
- Hood DA, Irrcher I, Ljubicic V, et al. Coordination of metabolic plasticity in skeletal muscle. J Exp Biol. 2006;209:2265–2275. - PubMed
-
- Muoio DM, Koves TR. Skeletal muscle adaptation to fatty acid depends on coordinated actions of the PPARs and PGC1 alpha: implications for metabolic disease. Appl Physiol Nutr Metab. 2007;32:874–883. - PubMed
-
- Gilde AJ, Van Bilsen M. Peroxisome proliferator-activated receptors (PPARS): regulators of gene expression in heart and skeletal muscle. Acta Physiol Scand. 2003;178:425–434. - PubMed
-
- Calvo JA, Daniels TG, Wang X, et al. Muscle-specific expression of PPARgamma coactivator-1alpha improves exercise performance and increases peak oxygen uptake. J Appl Physiol. 2008;104:1304–1312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

