Phosphatidylinositol 3-kinase/Akt pathway targets acetylation of Smad3 through Smad3/CREB-binding protein interaction: contribution to transforming growth factor beta1-induced Epstein-Barr virus reactivation
- PMID: 19589780
- PMCID: PMC2781985
- DOI: 10.1074/jbc.M109.036483
Phosphatidylinositol 3-kinase/Akt pathway targets acetylation of Smad3 through Smad3/CREB-binding protein interaction: contribution to transforming growth factor beta1-induced Epstein-Barr virus reactivation
Abstract
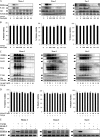
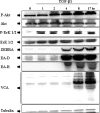
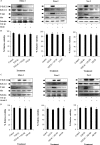
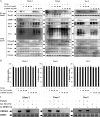
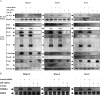
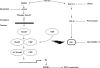
Epstein-Barr virus, a ubiquitous human herpesvirus, is associated with the development of carcinomas and lymphomas. We previously showed that transforming growth factor beta1 (TGF-beta1) mediated the virus to enter the lytic cycle, which is triggered by expression of Z Epstein-Barr virus replication activator (ZEBRA), through the ERK 1/2 MAPK signaling pathway. We report here that Akt, activated downstream from ERK 1/2, was required for TGF-beta1-induced ZEBRA expression and enabled Smad3, a mediator of TGF-beta1 signaling, to be acetylated by direct interaction with the co-activator CREB-binding protein and then to regulate TGF-beta1-induced ZEBRA expression.
Figures




Similar articles
-
Transforming growth factor beta 1 stimulates expression of the Epstein-Barr virus BZLF1 immediate-early gene product ZEBRA by an indirect mechanism which requires the MAPK kinase pathway.J Virol. 2000 Jul;74(13):5810-8. doi: 10.1128/jvi.74.13.5810-5818.2000. J Virol. 2000. PMID: 10846060 Free PMC article.
-
Induction of cardiac fibroblast lysyl oxidase by TGF-β1 requires PI3K/Akt, Smad3, and MAPK signaling.Cytokine. 2011 Jul;55(1):90-7. doi: 10.1016/j.cyto.2011.03.024. Epub 2011 Apr 17. Cytokine. 2011. PMID: 21498085
-
NF-kappaB-mediated modulation of inducible nitric oxide synthase activity controls induction of the Epstein-Barr virus productive cycle by transforming growth factor beta 1.J Virol. 2011 Jul;85(13):6502-12. doi: 10.1128/JVI.02560-10. Epub 2011 Apr 20. J Virol. 2011. PMID: 21507981 Free PMC article.
-
(-)-Epigallocatechin-3-gallate inhibition of Epstein-Barr virus spontaneous lytic infection involves ERK1/2 and PI3-K/Akt signaling in EBV-positive cells.Carcinogenesis. 2013 Mar;34(3):627-37. doi: 10.1093/carcin/bgs364. Epub 2012 Nov 24. Carcinogenesis. 2013. PMID: 23180656
-
Insulin-like growth factor-I inhibits transcriptional responses of transforming growth factor-beta by phosphatidylinositol 3-kinase/Akt-dependent suppression of the activation of Smad3 but not Smad2.J Biol Chem. 2003 Oct 3;278(40):38342-51. doi: 10.1074/jbc.M304583200. Epub 2003 Jul 21. J Biol Chem. 2003. PMID: 12876289
Cited by
-
Clinical Correlations of Polycomb Repressive Complex 2 in Different Tumor Types.Cancers (Basel). 2021 Jun 24;13(13):3155. doi: 10.3390/cancers13133155. Cancers (Basel). 2021. PMID: 34202528 Free PMC article.
-
TGF-β1 mediates the effects of aspirin on colonic tumor cell proliferation and apoptosis.Oncol Lett. 2018 Apr;15(4):5903-5909. doi: 10.3892/ol.2018.8047. Epub 2018 Feb 14. Oncol Lett. 2018. PMID: 29552221 Free PMC article.
-
Systems biology approach reveals genome to phenome correlation in type 2 diabetes.PLoS One. 2013;8(1):e53522. doi: 10.1371/journal.pone.0053522. Epub 2013 Jan 7. PLoS One. 2013. PMID: 23308243 Free PMC article.
-
Membrane type 1-matrix metalloproteinase/Akt signaling axis modulates TNF-α-induced procoagulant activity and apoptosis in endothelial cells.PLoS One. 2014 Aug 27;9(8):e105697. doi: 10.1371/journal.pone.0105697. eCollection 2014. PLoS One. 2014. PMID: 25162582 Free PMC article.
-
The role of PI3K/Akt in human herpesvirus infection: From the bench to the bedside.Virology. 2015 May;479-480:568-77. doi: 10.1016/j.virol.2015.02.040. Epub 2015 Mar 20. Virology. 2015. PMID: 25798530 Free PMC article. Review.
References
-
- Desgranges C., de-The G., Wolf H., zur Hausen H. (1975) IARC Sci. Publ. 11, 191–193 - PubMed
-
- Henle W., Henle G., Niederman J. C., Klemola E., Haltia K. (1971) J. Infect. Dis. 124, 58–67 - PubMed
-
- Pallesen G., Hamilton-Dutoit S. J., Rowe M., Young L. S. (1991) Lancet 337, 320–322 - PubMed
-
- Kieff (2007) Epstein-Barr Virus and Its Replication, Lippincott William & Wilkins, Philadelphia, PA
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

