The Akt kinases: isoform specificity in metabolism and cancer
- PMID: 19597332
- PMCID: PMC2997486
- DOI: 10.4161/cc.8.16.9335
The Akt kinases: isoform specificity in metabolism and cancer
Abstract
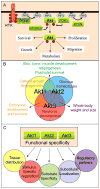
The Akt (PKB) protein kinases are critical regulators of human physiology that control an impressive array of diverse cellular functions, including the modulation of growth, survival, proliferation and metabolism. The Akt kinase family is comprised of three highly homologous isoforms: Akt1 (PKBalpha), Akt2 (PKBbeta) and Akt3 (PKBgamma). Phenotypic analyses of Akt isoform knockout mice documented Akt isoform specific functions in the regulation of cellular growth, glucose homeostasis and neuronal development. Those studies establish that the functions of the different Akt kinases are not completely overlapping and that isoform-specific signaling contributes to the diversity of Akt activities. However, despite these important advances, a thorough understanding about the specific roles of Akt family members and the molecular mechanisms that determine Akt isoform functional specificity will be essential to elucidate the complexity of Akt regulated cellular processes and how Akt isoform-specific deregulation might contribute to disease states. Here, we summarize recent advances in understanding the roles of Akt isoforms in the regulation of metabolism and cancer, and possible mechanisms contributing to Akt isoform functional specificity.
Figures

References
-
- Hanada M, Feng J, Hemmings BA. Structure, regulation and function of PKB/AKT--a major therapeutic target. Biochim Biophys Acta. 2004;1697:3–16. - PubMed
-
- Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, et al. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol. 1997;7:261–9. - PubMed
-
- Sarbassov DD, Guertin DA, Ali SM, Sabatini DM. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science. 2005;307:1098–101. - PubMed
-
- Bellacosa A, Chan TO, Ahmed NN, Datta K, Malstrom S, Stokoe D, et al. Akt activation by growth factors is a multiple-step process: the role of the PH domain. Oncogene. 1998;17:313–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
